Anthocyanin
Anthocyanin is a naturally occurring pH indicator that behaves similarly to litmus. It is actually a whole family of related organic compounds; the most useful particular anthocyanins are cyanidin (found in red cabbage, red wine) and chrysanthemin (found in hibiscus). Cyanidin is better soluble in water, chrysanthemin in ethanol, but their indicator properties are identical.
Contents
[hide]Properties
Physical
Anthocyanins are solid dye-like substances, not very stable. They are usually stored as solutions (chrysanthemin, dissolved in ethanol, is the best for long-term storage).
Chemical
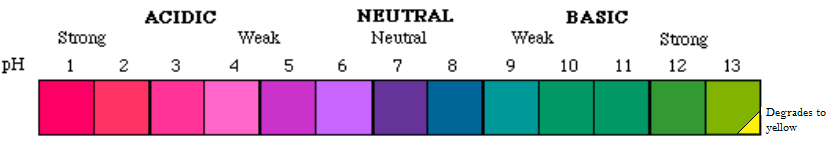
Cyanidin and chrysanthemin are useful pH indicators, with properties similar to litmus. In neutral pH, they are violet-blue. In low pH, they are red (very pronounced red in strongly acidic solutions, pink in weakly acidic ones). In high pH, they are green (bluish-green in weakly basic solutions, yellowish-green in strongly basic ones). In addition, extremely strong bases cause anthocyanins to slowly degrade, turn yellow and lose their indicator properties; concentrated alkalis first turn anthocyanins sickly green as normal, then in seconds or minutes they turn yellow.
Aqueous solutions of anthocyanins aren't terribly stable and degrade in a matter of days. Ethanol solutions of chrysanthemin are stable.
Availability
Anthocyanins can be extracted from common food products. Cyanidin is usually leached with water from red cabbage. The more desirable anthocyanin, chrysanthemin, is obtained from hibiscus herbal tea, which is marketed under names such as "Sorrel", "Red Zinger", "Karkade", "Italian Tea", "Flor de Jamaica", etc. in various countries.
Preparation
Cyanidin is usually prepared in the form of cabbage water, by infusing water on red cabbage leaves. Such an indicator is useful but inferior to chrysanthemin, which is leached by ethanol from hibiscus herbal tea. Do not use isopropanol: while it does dissolve chrysanthemin, the solubility is significantly lower and you won't have any deep or bright colors.
Once an anthocyanin solution is prepared, one has to adjust its pH. Cabbage water usually does not need such an adjustment, it is already purple. Hibiscus, on the other hand, also contains acids which make its extract reddish-pink. To adjust the pH of the alcohol solution, one should drop a basic solution such as sodium hydroxide or ammonia into it until it becomes purple-blue. Important: before you adjust the pH, filter the infusion off the hibiscus plant matter. Acids that the plant matter contains neutralize the base and ruin your work!
You can keep the dissolved form of anthocyanin, but test paper strips can also be made from it. In this, chrysanthemin is also superior: the alcoholic solution dries on paper much faster and gives a more uniform color. The best paper for the strips is filter paper, it gives the brightest colors with concentrated solutions. It is recommended to drench paper strips in the solution while it is still acidic, then neutralize it: the neutral form of anthocyanin is sparsely soluble in ethanol and forms a colloid, so the neutralizing agent acts as a mordant and results in brighter color of the strips.
You can also make acidic and basic test paper in addition to the universal one. This is recommended if you use normal paper for strips, which gives more pastel colors. Acidic test paper is pink or red rather than violet, it shows no color change with acids, but lets you identify bases easier. Basic test paper is green rather than violet, it shows no color change in bases (other than the yellow discoloration in very strong alkali) but lets you identify acids easier. Acidic test paper is easier to make: just skip the step of pH adjustment with chrysanthemin. To make basic paper, drop more base into the solution until it's green. However, do not overdo it: if your indicator solution or paper turns yellow, it is ruined and no longer good! That's why ammonia is preferable to nonvolatile bases for adjusting pH: when the test strips dry, a nonvolatile alkali can get too concentrated on the paper and ruin it, making it yellow and non-functional.
One package of herbal tea or one head of cabbage is enough for hundreds, maybe a thousand, of test paper strips. This makes this "DIY litmus" essentially free.
Projects
- Make DIY litmus substitute paper strips! Bajillions and metric craptons of paper strips! Free pH testing strips for everyone!
Handling
Safety
Dear Cthulhu forbid, what safety? Anthocyanins are edible!
Storage
Aqueous solutions of anthocyanins are very perishable. However, ethanol solutions and test paper have a long shelf life and do not require any special precautions to store.
Disposal
No special disposal is required. Any form of anthocyanin can be disposed of just like common garbage, into the trash bin or down the drain.