Difference between revisions of "Hydrazoic acid"
(pKa 4.7 is low-enough for it to qualify as mid-strength. It's the same as formic.) |
(→Relevant Sciencemadness threads) |
||
| (4 intermediate revisions by the same user not shown) | |||
| Line 61: | Line 61: | ||
| MeltingPt_ref = | | MeltingPt_ref = | ||
| MeltingPt_notes = | | MeltingPt_notes = | ||
| + | | Odor = Intolerable pungent odor | ||
| pKa = 4.6 <ref>Pradyot Patnaik. ''Handbook of Inorganic Chemicals''. McGraw-Hill, 2002, ISBN 0-07-049439-8</ref> | | pKa = 4.6 <ref>Pradyot Patnaik. ''Handbook of Inorganic Chemicals''. McGraw-Hill, 2002, ISBN 0-07-049439-8</ref> | ||
| pKb = | | pKb = | ||
| Solubility = Soluble | | Solubility = Soluble | ||
| − | | SolubleOther = | + | | SolubleOther = Reacts with alkali, [[amine]]s<br>Soluble in [[alcohol]], [[ether]]s |
| Solvent = | | Solvent = | ||
| VaporPressure = | | VaporPressure = | ||
| Line 139: | Line 140: | ||
==Projects== | ==Projects== | ||
*Synthesis of 2-Furonitrile | *Synthesis of 2-Furonitrile | ||
| − | *[[Silver azide|Silver]], [[lead azide|lead]] and [[mercury azide]] preparation | + | *[[Silver azide|Silver]], [[lead(II) azide|lead]] and [[mercury azide]] preparation |
*Preparation of azides that hydrolyze in aqueous solutions, such as magnesium or antimony azide (should only be performed in aprotic solvents that do not react with hydrazoic acid, or with extremely small amounts of pure cold acid and metal or none at all) | *Preparation of azides that hydrolyze in aqueous solutions, such as magnesium or antimony azide (should only be performed in aprotic solvents that do not react with hydrazoic acid, or with extremely small amounts of pure cold acid and metal or none at all) | ||
| Line 153: | Line 154: | ||
===Disposal=== | ===Disposal=== | ||
Hydrazoic acid can be neutralized with an excess of sodium hydroxide. However, azide salts such as the sodium azide produced in this way are extremely toxic, and they should NEVER be disposed of as is, especially not down the drain. Sodium azide produced this way should either be destroyed with [[nitrous acid]], crystallized out by evaporation and carefully heated to decomposition outside, or precipitated as a transition metal azide. Many transition metal azides are explosive or will hydrolyze, and these too must be very carefully disposed of. | Hydrazoic acid can be neutralized with an excess of sodium hydroxide. However, azide salts such as the sodium azide produced in this way are extremely toxic, and they should NEVER be disposed of as is, especially not down the drain. Sodium azide produced this way should either be destroyed with [[nitrous acid]], crystallized out by evaporation and carefully heated to decomposition outside, or precipitated as a transition metal azide. Many transition metal azides are explosive or will hydrolyze, and these too must be very carefully disposed of. | ||
| + | |||
| + | Bleach (dil. sodium hypochlorite) can also be used.<ref>https://www.ncbi.nlm.nih.gov/m/pubmed/20667654/</ref> | ||
==References== | ==References== | ||
| Line 159: | Line 162: | ||
*[http://www.sciencemadness.org/talk/viewthread.php?tid=1987 Azides] | *[http://www.sciencemadness.org/talk/viewthread.php?tid=1987 Azides] | ||
*[http://www.sciencemadness.org/talk/viewthread.php?tid=27640 The true mad science, hydrogen azide distillation] | *[http://www.sciencemadness.org/talk/viewthread.php?tid=27640 The true mad science, hydrogen azide distillation] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=156715 Has anyone smelled hydrazoic acid?How does it smell like?Is it worse than chlorine,ozone and nitrogen dioxide?] | ||
[[Category:Chemical compounds]] | [[Category:Chemical compounds]] | ||
| Line 171: | Line 175: | ||
[[Category:Things that can kill you very quickly]] | [[Category:Things that can kill you very quickly]] | ||
[[Category:Blood agents]] | [[Category:Blood agents]] | ||
| + | [[Category:Liquids]] | ||
Latest revision as of 22:42, 13 February 2021
| Names | |
|---|---|
| IUPAC name
Hydrogen azide
| |
| Other names
Azoimide
Hydrogen azide | |
| Identifiers | |
| Jmol-3D images | Image |
| |
| Properties | |
| HN3 | |
| Molar mass | 43.03 g/mol |
| Appearance | Colorless liquid |
| Odor | Intolerable pungent odor |
| Density | 1.09 g/cm3 |
| Melting point | −80 °C (−112 °F; 193 K) |
| Boiling point | 37 °C (99 °F; 310 K) |
| Soluble | |
| Solubility | Reacts with alkali, amines Soluble in alcohol, ethers |
| Acidity (pKa) | 4.6 [1] |
| Thermochemistry | |
| Std enthalpy of
formation (ΔfH |
300.25 kJ/mol |
| Hazards | |
| Safety data sheet | Guidechem |
| Related compounds | |
| Related compounds
|
Ammonia Hydrazine Triazane |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Hydrazoic acid, also known as hydrogen azide or azoimide, is a colorless, volatile, and explosive liquid at room temperature and pressure, with the chemical formula HN3.
Contents
Properties
Chemical
Hydrazoic acid is a mid-strength acid, it will dissolve many metals, such as iron, copper, aluminium, magnesium, zinc. Antimony dissolves slowly in hydrogen azide, as does silver at high concentrations. When mixed with hydrochloric acid, it can attack gold and platinum. Platinum black will decompose hydrogen azide to ammonia and nitrogen.[2]
Dissolution in the strongest acids produces explosive salts containing the H2N=N=N+ ion, for example:
- HN=N=N + HSbCl6 → [H2N=N=N+] + [SbCl6]−
Hydrazoic acid decomposes to nitrogen and hydrogen:
- 2HN3 → H2 + 3N2
Physical
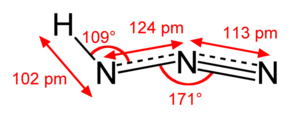
Hydrazoic acid is a colorless liquid, with a strong and very unpleasant smell. It is very soluble in water, as well as alcohol and ether. Its density is 1.09 g/cm3. Anhydrous hydrozoic acid melts at −80 °C and boils at 37 °C. It is a mid-strength acid (pKa = 4.75), of approximately the same strength as formic acid.
Availability
Due to its long list of hazards, hydrazoic acid is not sold, and has to be prepared in the laboratory.
Preparation
Hydrazoic acid can be prepared by reacting a strong acid, such as sulfuric acid, with an azide, such as sodium azide or barium azide. The latter is preferred as the insoluble barium sulfate produced can simply be filtered out, giving a clean solution of hydrogen azide. It's recommended to produce diluted acid, as pure hydrazoic acid is prone to detonation.
Hydrazoic acid was originally prepared by the reaction of hydrazine with nitrous acid:
- N2H4+ HNO2 → HN3 + 2 H2O
As nitrous acid is already diluted, this reaction is safer. It can similarly be accomplished using nitric acid or concentrated hydrogen peroxide.
Projects
- Synthesis of 2-Furonitrile
- Silver, lead and mercury azide preparation
- Preparation of azides that hydrolyze in aqueous solutions, such as magnesium or antimony azide (should only be performed in aprotic solvents that do not react with hydrazoic acid, or with extremely small amounts of pure cold acid and metal or none at all)
Handling
Safety
Hydrazoic acid is volatile and highly toxic, similar in toxicity with cyanides, however it has no known antidote. It has a pungent smell and its vapors can cause strong headaches. In pure form it is prone to detonation, so it's best to work with diluted solutions. Azide salts carry the same dangers, and are prone to hydrolysis in aqueous solution.
In case of hydrazoic acid poisoning, the patient should immediately leave the contaminated room (or be carried from there) and given access to outside air. In case of severe poisoning, the patient should immediately receive medical attention. If no medical attention is available, the patient should be given pure oxygen to breathe.
Storage
Hydrazoic acid should only be stored as diluted solutions, is closed bottles, in well ventilated areas or in a special storage cabinet. Should not be stored for long periods of time.
Disposal
Hydrazoic acid can be neutralized with an excess of sodium hydroxide. However, azide salts such as the sodium azide produced in this way are extremely toxic, and they should NEVER be disposed of as is, especially not down the drain. Sodium azide produced this way should either be destroyed with nitrous acid, crystallized out by evaporation and carefully heated to decomposition outside, or precipitated as a transition metal azide. Many transition metal azides are explosive or will hydrolyze, and these too must be very carefully disposed of.
Bleach (dil. sodium hypochlorite) can also be used.[3]
References
- ↑ Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0-07-049439-8
- ↑ Robert Matyáš, Jiří Pachman, Primary Explosives, 2013, Chapter 4 Azides
- ↑ https://www.ncbi.nlm.nih.gov/m/pubmed/20667654/
Relevant Sciencemadness threads
- Chemical pages without CAS Registry Number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Chembox articles without image
- Chemical compounds
- Inorganic compounds
- Hydrogen compounds
- Nitrogen compounds
- Acids
- Mid-strength acids
- Mineral acids
- Azides
- Energetic materials
- Things that can kill you very quickly
- Blood agents
- Liquids