Nitric acid
| |
| Names | |
|---|---|
| IUPAC name
Nitric acid
| |
| Systematic IUPAC name
Nitric acid | |
| Other names
Acidum nitricum
Aqua fortis Eau forte Hydrogen nitrate Spirit of niter | |
| Properties | |
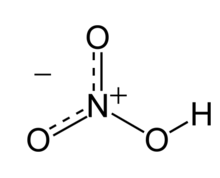
| HNO3 | |
| Molar mass | 63.01 g/mol |
| Appearance | Colorless, yellow or red fuming liquid |
| Odor | Acrid, acidic |
| Density | 1.5129 g/cm3 |
| Melting point | −42 °C (−44 °F; 231 K) |
| Boiling point | 83 °C (181 °F; 356 K) 121 °C (68%) |
| Miscible | |
| Vapor pressure | 48 mmHg (20 °C) |
| Acidity (pKa) | -1.4 |
| Thermochemistry | |
| Std molar
entropy (S |
146 J·mol−1·K−1 |
| Std enthalpy of
formation (ΔfH |
−207 kJ/mol |
| Hazards | |
| Safety data sheet | Sigma-Aldrich (anhydrous) Sigma-Aldrich (azeotrope) |
| Flash point | Non-flammable |
| Related compounds | |
| Related compounds
|
Nitrous acid |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Nitric acid is a strong acid with the formula HNO3. It is an important mineral acid, along with hydrochloric, sulfuric, perchloric, and phosphoric acids. It is a powerful oxidizing agent, especially when mixed with sulfuric acid, which produces the nitronium ion in situ.
Contents
Properties
Chemical
Nitric acid is an oxidizing acid at room temperature. It is often used in the nitration of organic compounds. It is capable of dissolving metals such as copper and silver due to its oxidizing nature, and will release toxic nitrogen dioxide as an oxidation by product.
- Cu + 8 HNO3 → 3 Cu(NO3)2 + 2 NO + 4 H2O
Upon reaction with nitric acid, most metals give the corresponding nitrates. Some metalloids and metals give the oxides instead. For example, tin, arsenic, antimony and titanium are oxidized into tin(IV) oxide, arsenic oxide, antimony oxide, and titanium dioxide respectively. Concentrated nitric acid will form a protective layer on the surfaces of some metals, like iron, cobalt, chromium, nickel, and aluminium, which prevents the metal from further reacting with the nitric acid.
Some precious metals, such as pure gold and platinum group metals do not react with nitric acid, though pure gold and platinum do react with aqua regia, a mixture of concentrated nitric and hydrochloric acids.
Concentrated nitric acid oxidizes elemental iodine, phosphorus and sulfur into iodic acid, phosphoric acid and sulfuric acid respectively. Although it reacts with graphite and amorphous carbon and graphite, yielding carbon dioxide, it does not react with diamond. This property can be used to separate diamond from the graphite. Elemental boron resists nitric acid at standard conditions, but hot nitric acid will slowly oxidize it to boric acid. It will also not react with nitrogen, oxygen, noble gases, silicon and the first three halogens (fluorine, chlorine, bromine).
Nitric acid is known to slowly decompose over time, a process more visible in conc. acid:
- 4 HNO3 → 2 H2O + 4 NO2 + O2
Nitration of organic compounds with nitric acid is the primary method of synthesis of many common explosives and other energetic materials, such as nitroglycerin, nitrocellulose, trinitrotoluene, picric acid, RDX, etc. Conc. sulfuric acid is used to aid the nitration.
Nitric acid reacts with proteins to form yellow nitrated products. This reaction is known as the xanthoproteic reaction and it's used to detect the presence of proteins in a solution. The test is carried out by adding concentrated nitric acid to the substance being tested, and then heating the mixture. If proteins that contain amino acids with aromatic rings are present, the mixture turns yellow. Upon adding a base such as ammonia, the color turns orange. These color changes are caused by nitrated aromatic rings in the protein.
Physical
Concentrated nitric acid is a clear solution with a density of about 1.2 g/ml. When the concentration of the acid surpasses 70%, it becomes classified as fuming nitric acid, this can be identified by visible fuming when air is blown into it. Nitric acid in concentrations above 90% fumes extremely profusely on any contact with air. It forms an azeotrope with water at 68% concentration, which makes it difficult to produce the pure substance.
Availability
Nitric acid is available from lab suppliers like Elemental Scientific and Duda Diesel. While the acid itself is not expensive, it requires a mandatory HAZMAT shipping fee of $37.50, making this acid rather expensive for the amateur chemist.
Most places limit the sale of nitric acid to the general public, due to its use in the manufacturing of explosive materials. In the EU, the sale and possession of nitric acid in conc. higher than 3% (yes, THREE PERCENT) is forbidden without a license. Conc. nitric acid is also restricted to the general public in other countries.
Preparation
The classic method of laboratory synthesis of nitric acid is described in a sub-article:
Glauber's nitric acid synthesis
A somewhat less efficient method of producing nitric acid is by the reaction of a mixture containing sulfuric acid and a nitrate salt with copper metal, generating large amounts of nitrogen dioxide gas. The gas may then be bubbled into hydrogen peroxide or water, with hydrogen peroxide producing a higher yield.
Sodium bisulfate can also be use to replace sulfuric acid, however the higher temperature will decompose more of the nitric acid to oxygen, nitrogen dioxide and water.
It is possible to produce nitric acid with an Ostwald reactor, or by reacting nitrogen and oxygen in air with an electric spark.
If you need to quickly whip up some dilute nitric acid, you can use the "quick and dirty" method, namely reacting calcium nitrate with sulfuric acid. This method has two downsides: first, even if your reagents are in a perfectly stoichiometric ratio, the resulting acid is still contaminated by a small amount of calcium sulfate that managed to stay in solution. Second, the precipitate of calcium sulfate is incredibly messy and voluminous, the acid looks like sour cream and it is almost impossible to decant it properly. Use this method only if you have a vacuum filtering set. This low-grade acid can be distilled to yield azeotropic nitric acid.
To get past azeotropic nitric acid and obtain red fuming nitrate acid, the azeotropic acid is distilled in the presence of a drying agent, such as solid magnesium nitrate or concentrated (>90%) sulfuric acid.
Yellow concentrated nitric acid can be turned white, or RFNA converted to WFNA, by bubbling oxygen though the acid. Oxygen oxidizes NO2 to N2O5, which combines with the residual water in your acid to produce nearly pure nitric acid. Vacuum drying will also remove the NO2 gas.
Projects
Nitric acid can be used for many projects including make nitrate salts. When mixed with concentrated sulfuric acid or hydrofluoric acid, nitric acid acts like a base and releases a nitronium ion:
- 2 H2SO4 + HNO3 → NO2+ + 2 HSO4- + H2O
This mixture, known as nitrating mixture or mixed acid can be used for nitrating many organic compounds.
Other:
- Make oxalic acid
- Make aqua regia
- Purify silver
- Make nitrated explosive compounds
- Artificially age wood (pine and maple)
- Make nital (etchant)
- Pickling stainless steel
Handling
Safety
Nitric acid solutions are highly corrosive and will stain the skin yellow as the proteins are nitrated. Care should be taken to not let nitric acid contact the skin. Nitrates should not be used with hydrochloric acid, because this generates nitrosyl chloride.
Most glove types (with the notable exceptions butyl rubber or neoprene) are incompatible due to the strong oxidizing effects of nitric acid and may burn upon contact with the acid.[1] When handling fuming nitric acid, wear butyl rubber gloves. Other types of rubber may react extremely violently with nitric acid of this concentration. If you don't have butyl rubber, don't wear gloves at all: damage done to your bare hands will be lesser than what rubber on fire can do to you. Nitric acid in general reacts with rubber; do not try to distill it in apparata that contain rubber parts. Use ground glass joints or a retort when distilling nitric acid, especially of fuming concentration.
Storage
Nitric acid is incompatible with most plastics due to its oxidizing nature, though bottle lids made of polypropylene (PP) are acceptable. High concentrations of nitric acid are light sensitive and should be kept in amber glass bottles with ample headroom to prevent pressure build up from nitrogen oxides.
Disposal
Nitric acid can be neutralized with neutralizing compounds, such as carbonates, bicarbonates, oxides, hydroxides. Calcium carbonate is a good neutralizing agent, and, as long as the acid isn't contaminated with heavy metals, the resulting calcium nitrate can be discarded in ground or poured down the drain. Concentrated nitric acid (>50%) must first be diluted in cold water, before being neutralized with a base, to limit the amount of corrosive fumes/aerosols released in air during the neutralization process.
