Jackson
Hazard to Others
  
Posts: 189
Registered: 22-5-2018
Location: U S of A
Member Is Offline
Mood:  Happy about new glassware 
|
|
Questions about azeotropes?
In the case of an azeotropic ethanol, if its concentration percentage was raised with anhydrous ethanol to say 98% and then distilled, would all of it
come over as 98% or would it come over as 100% at the beginning and then azeotropic afterward?
|
|
|
oberkarteufel
Harmless

Posts: 47
Registered: 11-12-2018
Member Is Offline
Mood: Mayonesium sulfate
|
|
Azeotrope has a lower boiling point, so it will distill off first, leaving solution with higher ethanol content.
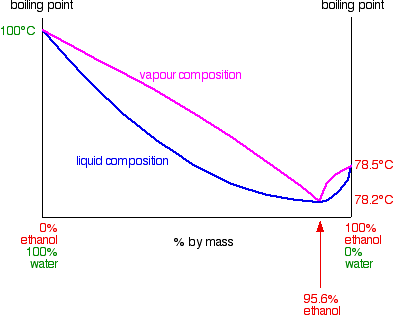
|
|
|
DavidJR
National Hazard
   
Posts: 908
Registered: 1-1-2018
Location: Scotland
Member Is Offline
Mood: Tired
|
|
I've posted about this before:
Quote: Originally posted by DavidJR  | Example 1: Zeotropic mixtures
Consider a mixture of methanol (b.p. 65c) and water (b.p. 100c), which do not form an azeotrope. You can be certain that the boiling point of any such
mixture is between the boiling points of the components, which covers the full range of %methanol. More importantly, the vapour produced by boiling
such a mixture will always be richer in the lower-boiling component (methanol) than the mixture you started with. This means you can easily separate
them by fractional distillation.
Example 2: Minimum-boiling (positive) azeotropic mixtures
Consider a mixture of isopropanol (b.p. 83c) and water (b.p. 100c). Isopropanol and water form an azeotrope at 91% v/v isopropanol, which boils at
~80c.
2a: If our mixture contains 50% v/v isopropanol and we put it in a simple distillation apparatus, with the boiling flask heated by a
water/oil/sand bath at 85c, the mixture will boil producing a vapour richer in isopropanol than the starting mixture. Once the mixture stops boiling
(since temp only 85c we won't drive over all of the water) we can empty out the boiling flask, and put the distillate in the boiling flask for a
second run. This time we'll still increase the % isopropanol in the distillate, but not by as much as we did before. We can repeat this, but with
diminishing returns. Essentially a fractionating column would do this work for you as the vapour has the opportunity to recondense/reevaporate many
times. The most concentrated you'll ever get is 91% v/v because at this point we have an azeotrope, in other words, the %isopropanol in the vapour
produced by boiling the mixture is the same as in the liquid.
2b: If instead our starting mixture contained 95% isopropanol (above the azeotrope) then boiling this would produce vapour containing less
isopropanol (though still above the azeotrope). So, if we were to fractionally distill our 95% IPA, the distillate would be the 91% azeotrope.
However, if we distill until all of the water has been carried over as the azeotrope, then the residue left in the boiling flask will be ~100%
isopropanol.
Example 3: Maximum-boiling (negative) azeotropic mixtures
Consider a mixture of nitric acid (b.p. 83c) and water (b.p. 100c). Nitric acid and water form an azeotrope at 68% w/w HNO3, which boils at
121c.
3a: If our mixture contains 50% w/w HNO3, then we have more water than the azeotrope. So if we perform a fractional distillation,
then the excess water will come over first at 100c, and then the azeotrope will come over at 121c.
3b: If instead our starting mixture contained 85% w/w HNO3, then we would have less water than the azeotrope. So if we perform a
fractional distillation, then the excess HNO3 will come over first at 83c, and after that, the azeotrope will come over at 121c.
N.B. for simplicity of explanation i've assumed in these examples that your fractionating column is impossibly good
|
See example 2b.
[Edited on 31-1-2019 by DavidJR]
[Edited on 31-1-2019 by DavidJR]
|
|
|
Jackson
Hazard to Others
  
Posts: 189
Registered: 22-5-2018
Location: U S of A
Member Is Offline
Mood:  Happy about new glassware 
|
|
Thanks for all the replies. I just want to make sure that I have a good understanding of the concepts.
|
|
|