HydroCarbon
Hazard to Self
 
Posts: 77
Registered: 7-7-2008
Location: Anytown, USA
Member Is Offline
Mood: No Mood
|
|
Need Help with a Project ( Polymer Chemistry)
I have an assignment to design a polymer that could be used for water purification. My idea is for a polymer that has a structure similar to EDTA
that can chelate harmful metal ions like Pb Hg and Cd. The monomer structure I have drawn out is this
CH2=CHNHCH2CO2Na
(CO2Na is an acid group with sodium counter ion)
The molecule will then go through free radical polymerization. The product will be much like polystyrene except the pendant groups will be
-NHCH2CO2Na, which can chelate the metals, rather than the aromatic rings in ps.
So here are the questions: Does this monomer even exist? if so how would it be named? I tried searching couldn't find anything
Also, if it does exist, will it polymerize? at this point I'm worried the pendant groups might be too bulky and have too much charge based repulsion.
Any help is much appreciated.
|
|
|
HydroCarbon
Hazard to Self
 
Posts: 77
Registered: 7-7-2008
Location: Anytown, USA
Member Is Offline
Mood: No Mood
|
|
Ok, new addition to the plan. I now have decided to make the reaction 40% ethylene and 60% of the monomer above. That way there will be polyethylene
units between the chelating pendants, therefore reducing repulsion between the pendant groups, but hopefully not affecting chelating properties.
[Edited on 1-12-2009 by HydroCarbon]
|
|
|
not_important
International Hazard
    
Posts: 3873
Registered: 21-7-2006
Member Is Offline
Mood: No Mood
|
|
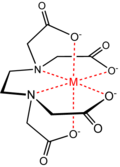
I suggest that you look at the attached image of an EDTA chelate structure, then ask if similar can be formed by your polymer.
Nitrilotriacetic acid is a vaguely similar chelating agent. Citric acid and polyphosphates are also chelators.
For some heavier metals sulfur based compounds (thiols) work better than the amine based ones. An example is given by DOI: 10.1002/jps.2600710919 -
Chelation of mercury by polymercaptal microspheres: New potential antidote for mercury poisoning
|
|
|
watson.fawkes
International Hazard
    
Posts: 2793
Registered: 16-8-2008
Member Is Offline
Mood: No Mood
|
|
You might look at polyvinylpyrrolidone, the polymer that holds iodine in povidone-iodine. It's not chelating a metal, but you can get some insight as
to a geometry that does the kind of complexing you're looking for.
|
|
|