| Pages:
1
..
47
48
49
50
51
..
66 |
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
You probably are: there just isn't enough t-butanol to consume much K. To be precise, even if your conversion was 100 %, you'd still only get 90 % of
the theoretical K because out each 1 mol of actual, dry KOH 0.1 mol gets tied up as K t-BuO at the end of the reaction. It's all explained higher up
in much detail. During all of the reaction, including post-reaction, 0.1 mol of each mol of K is 'tied up' as K t-BuO. But 0.1 mol is only 10 % of the
total quantity of K (if the KOH/cat. ratio is 1 to 0.1 mol)
No, if you had K forming and then disappearing the most likely explanation is an 'outside' scavenger to reoxidise the K. Oxygen, water or 'something'
in the solvent...
[Edited on 10-3-2013 by blogfast25]
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
If you believe you've failed, at least subject a sample of the gunge to water. ANY K in there will reveal itself quickly.
It could also be useful to subject a sample of the gunge to strong HCl to check solubility and MgO. Then separate off the watery phase and check for
magnesium: magnesium carbonate is poorly soluble in water, so after neutralisation you should get some precipitation with Na2CO3.
Mailinmypocket, what temperature control/monitoring do you have in place? Going by your last pictures I see everything is fine but not a hint of
thermometer. That's a serious limitation. There's room for a third, small hole in that stopper, for a thermocouple probe. Household digithermometers
go up to 200 C, it's what I used.
[Edited on 10-3-2013 by blogfast25]
|
|
|
Mailinmypocket
International Hazard
    
Posts: 1351
Registered: 12-5-2011
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by blogfast25  | If you believe you've failed, at least subject a sample of the gunge to water. ANY K in there will reveal itself quickly.
It could also be useful to subject a sample of the gunge to strong HCl to check solubility and MgO. Then separate off the watery phase and check for
magnesium: magnesium carbonate is poorly soluble in water, so after neutralisation you should get some precipitation with Na2CO3.
Mailinmypocket, what temperature control/monitoring do you have in place? Going by your last pictures I see everything is fine but not a hint of
thermometer. That's a serious limitation. There's room for a third, small hole in that stopper, for a thermocouple probe. Household digithermometers
go up to 200 C, it's what I used.
[Edited on 10-3-2013 by blogfast25] |
I will test the residues later this afternoon following your suggested procedure. The temperature is indeed being monitored by an electronic kitchen
thermometer, it can be seen in the background. The probe was buried under the sand, but not directly on the bottom of the pot. The temp fluctuated
between 220-240 thanks to my shitty old stove, I would have used the hotplate but the Corning manual says to not heat sandbaths on it so I played it
safe. Ill report on the outcome of the gunge test later...
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
MIMP:
Blindness is a terrible affliction: I didn't see your nice probe there, my bad  .
That should really rule out any temperature problems. .
That should really rule out any temperature problems.
|
|
|
elementcollector1
International Hazard
    
Posts: 2684
Registered: 28-12-2011
Location: The Known Universe
Member Is Offline
Mood: Molten
|
|
I'm going to see if I can get a stronger heat source. What I have now is a Hamilton electric hot plate, and while it appears to reach 200 C, there's
no guarantee that that's true.
A propane burner should be more than good, correct?
Also, per the oxygen and water destroying the K: Is there any way to remove these two without going inside? Again, parents. If I could, I'd flush the
container with argon.
After 6 failures in a row, I'm beginning to lose hope for this experiment. I'll keep trying, and posting the results, though.
Elements Collected:52/87
Latest Acquired: Cl
Next in Line: Nd
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by elementcollector1  | I'm going to see if I can get a stronger heat source. What I have now is a Hamilton electric hot plate, and while it appears to reach 200 C, there's
no guarantee that that's true.
A propane burner should be more than good, correct?
Also, per the oxygen and water destroying the K: Is there any way to remove these two without going inside? Again, parents. If I could, I'd flush the
container with argon.
After 6 failures in a row, I'm beginning to lose hope for this experiment. I'll keep trying, and posting the results, though. |
Propane should be fine. I used it. Always with sand bath though.
If your plate does 200 C your flask will by definition be slightly cooler. You really should have a temperature probe poking into the reaction
chamber...
Argon shouldn't be necessary. If there is something oxidising the K it shouldn't be there in the first place and argon probably wouldn't get rid of
it. Maybe it really is time to try Shellsol D?
|
|
|
hyfalcon
International Hazard
    
Posts: 1003
Registered: 29-3-2012
Member Is Offline
Mood: No Mood
|
|
If you can find a source please u2u me. I've searched and searched for this in the US and can't find it.
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
I'm not US based so I can't help you there. But higher up in this thread a US based poster did find it without too many problems. The patent is also a
US patent, I seem to recall...
|
|
|
elementcollector1
International Hazard
    
Posts: 2684
Registered: 28-12-2011
Location: The Known Universe
Member Is Offline
Mood: Molten
|
|
Say, could the lamp oil be dried with NaOH or some such? Sodium metal? (It would be a bit ironic, but still). Anhydrous MgSO4 is good, but these two
are a bit easier to prepare.
I'll see if I can pick up a lab thermometer or something from school today (hopefully it can do 200 C). Maybe then I can check the boiling point of
this stuff, and see how it matches. If the school is a no-go, then I'll probably get a meat thermometer from Target.
Elements Collected:52/87
Latest Acquired: Cl
Next in Line: Nd
|
|
|
Fantasma4500
International Hazard
    
Posts: 1681
Registered: 12-12-2012
Location: Dysrope (aka europe)
Member Is Offline
Mood: dangerously practical
|
|
i think ive found a person who can approve that its possible to melt potassium nitrate, lead electricity through this molten potassium nitrate and
without the formed K metal reacting explosively or at all with the KNO3..
he use KNO3 and KI to get iodine vapours in what i saw, but he said that when a drop of water is added to one of the cathodes (the one with potassium
ofcourse) it sparks violently
imagine turning magnesium salts into pure magnesium metal with electricity and abit of heat..
hope some of you guys will be as hopeful towards this as i am.. because i am hoping that this could be possible.. it will open up for so many
possibilities and rare metals by electrolysis without +1000*C uncontrolled reactions
|
|
|
elementcollector1
International Hazard
    
Posts: 2684
Registered: 28-12-2011
Location: The Known Universe
Member Is Offline
Mood: Molten
|
|
I'm guessing that mixture melts at 500 C? It could work, but any potassium produced is immediately exposed to oxygen. Sodium has been done with NaOH,
however, by use of a homemade Castner cell ("Hot electrochemical sodium").
Molten electrolysis isn't exactly a new idea, it's just hard to accomplish effectively. That, and the reactivity of some metals such as K, makes this
catalytic synthesis more viable (or does it?).
Elements Collected:52/87
Latest Acquired: Cl
Next in Line: Nd
|
|
|
Fantasma4500
International Hazard
    
Posts: 1681
Registered: 12-12-2012
Location: Dysrope (aka europe)
Member Is Offline
Mood: dangerously practical
|
|
true.. but ive never seen it done more than once with nitrates, and using nitrates would be abit more smart as you dont have highly corrosive liquid
(which can etch glass!!)
also its usually easier to get a hold of a metal nitrate than the metal hydroxide (or well atleast easier to make the nitrate, especially when you got
10L HNO3)
usually nitrates have a very low melting point aswell..
|
|
|
elementcollector1
International Hazard
    
Posts: 2684
Registered: 28-12-2011
Location: The Known Universe
Member Is Offline
Mood: Molten
|
|
It's viable, but I'd be reluctant to play with a powerful oxidizer and electricity.
Hydroxides have an even lower melting point, but tend to be harder to store or use due to the reaction with glass, as you said.
On a side note, our school thermometers go to only 50 C. Pathetic! I'm going to Target ASAP for a meat thermometer.
Elements Collected:52/87
Latest Acquired: Cl
Next in Line: Nd
|
|
|
Fantasma4500
International Hazard
    
Posts: 1681
Registered: 12-12-2012
Location: Dysrope (aka europe)
Member Is Offline
Mood: dangerously practical
|
|
yes it might be potentially dangerous to play around with molten nitrates.. as it tends to ignite fuels on contact.. (:
the guy i saw do it used graphite electrode, tho.. (strange?)
but the idea is to be able to do it much more controlled than making a thermite and tonnes of tiny pieces of metal..
by electrolysis of lead bromide you get bromine (ofcourse) and lead, the more interesting in this is that it forms on one of the electrodes and then
falls to the bottom, which means you can break the solid lump up after its cooled down and you should be able to get a solid piece of metal, i wonder
if this works with potassium or sodium aswell, or if its too light to fall to the bottom..
|
|
|
elementcollector1
International Hazard
    
Posts: 2684
Registered: 28-12-2011
Location: The Known Universe
Member Is Offline
Mood: Molten
|
|
Again, read "hot electrochemical sodium". It's a stickied thread in Technochemistry, IIRC.
Elements Collected:52/87
Latest Acquired: Cl
Next in Line: Nd
|
|
|
Fantasma4500
International Hazard
    
Posts: 1681
Registered: 12-12-2012
Location: Dysrope (aka europe)
Member Is Offline
Mood: dangerously practical
|
|
really..?? well thanks.. havent heard of it before tho
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Downs Cell:
http://en.wikipedia.org/wiki/Downs_cell
|
|
|
elementcollector1
International Hazard
    
Posts: 2684
Registered: 28-12-2011
Location: The Known Universe
Member Is Offline
Mood: Molten
|
|
Anyway, back to business. I got a temperature probe with a range of -40 C to 230 C, and while it's not made for long, sustained readings, it seems to
work just fine. The first test I made for the BP of the lamp oil oddly resulted in the lamp oil never boiling, and getting stuck at 165 C. Maybe I'll
remove some sand from the bath and try that again.
Elements Collected:52/87
Latest Acquired: Cl
Next in Line: Nd
|
|
|
Mailinmypocket
International Hazard
    
Posts: 1351
Registered: 12-5-2011
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by blogfast25  | If you believe you've failed, at least subject a sample of the gunge to water. ANY K in there will reveal itself quickly.
It could also be useful to subject a sample of the gunge to strong HCl to check solubility and MgO. Then separate off the watery phase and check for
magnesium: magnesium carbonate is poorly soluble in water, so after neutralisation you should get some precipitation with Na2CO3.
Mailinmypocket, what temperature control/monitoring do you have in place? Going by your last pictures I see everything is fine but not a hint of
thermometer. That's a serious limitation. There's room for a third, small hole in that stopper, for a thermocouple probe. Household digithermometers
go up to 200 C, it's what I used.
[Edited on 10-3-2013 by blogfast25] |
Okay, got around to testing the residues and I'm thinking not much is going on as far as the reaction is concerned.
Image 1: Isolated white crust only, in the left tube. Magnesium bits in the right tube. I tested Mg just... Because.
Image 2: After adding approximately 15% HCl to both tubes, the white residue made a frightening cracking sound. I assume this is because it is simply
KOH which does not like being thrown into acid, it heated up substantially. The Mg reacted predictably, with violent bubbling and heat to the point of
steam production.
Image 3: After neutralization of the tube containing the dissolved residue with sodium carbonate, no precipitate materialized.
The Mg surfaces were dark- almost black, I don't think the size of the particles are small enough...
  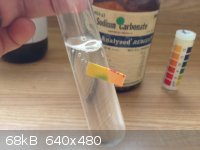
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by Mailinmypocket  | [Image 2: After adding approximately 15% HCl to both tubes, the white residue made a frightening cracking sound. I assume this is because it is simply
KOH which does not like being thrown into acid, it heated up substantially. The Mg reacted predictably, with violent bubbling and heat to the point of
steam production.
|
Yes, solid KOH + strong HCl means a lot of heat of neutralisation all at once.
You've more or less proved no reaction between the KOH and Mg took place. While particle size could be a factor, I seem to recall someone having
success with shavings instead of powder.
In this reduction reaction several steps occur, some of them require solid/liquid reactions, others do not. Without knowing what is the slowest step
in the chain it's hard to cast 'the blame' on any given factor.
But by all means try a finer grade of Mg and see what you get....
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by elementcollector1  | | Anyway, back to business. I got a temperature probe with a range of -40 C to 230 C, and while it's not made for long, sustained readings, it seems to
work just fine. The first test I made for the BP of the lamp oil oddly resulted in the lamp oil never boiling, and getting stuck at 165 C. Maybe I'll
remove some sand from the bath and try that again. |
You said the plate was up to 200 C. What you observe would fit that. With the plate at 200 C the flask can NEVER actually reach that temperature
because of insulation provided by the sand and heat losses of the flask to the environment. 165 C is quite different from 200 C. You'll either need a
hotter plate or switch to propane.
I think this is real progress for you, EC1...
[Edited on 13-3-2013 by blogfast25]
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Just to try and focus the attention a bit, I'll quote myself from here:
http://www.sciencemadness.org/talk/viewthread.php?tid=14970&...
Below is the simplest form of the supposed reaction path (it was later refined a bit).
Quote: Originally posted by blogfast25  |
It’s very clear from Example 1 (potassium from KOH) that the initial hydrogen comes from moisture reacting away (assuming for argument’s
sake that neither the patent nor pok are hoaxes). During that step no t-butanol is present yet.
The actual reactions in the second step could possibly be (with R a t-alkyl group):
Initiation:
KOH + ROH < --- > KOR + H2O
Wherever this equilibrium lies, it may be pulled to the right by:
Propagation:
2 KOR + Mg < --- > 2 K + Mg(OR)2
Which may itself by pulled to the right by:
Termination:
Mg(OR)2 + H2O --- > MgO + 2 ROH
Of the last step at least we can be reasonably sure: Mg alkoxides should be quite prone to hydrolysis.
2 KOH + Mg --- > 2 K + MgO + H2O
… would be the overall reaction in that scheme. A calculation using NIST values of HoF and S at 298 K, shows the ΔG = ΔH – TΔS to
be about -48.6 kJ/mole (of Mg reacted, @ 298 K) [edit: or about - 100 kJ/mole of K produced], so just barely thermodynamically possible. For higher
temperatures this requires a correction that is likely to be small. The low change in Gibbs free energy could explain low reaction rates.
[Edited on 4-12-2010 by blogfast25] |
Assuming this is at least conceptually correct, we still don’t know what is the rate limiting step. If the rate limiting step involves
solid magnesium, then fineness (total available surface area) would almost certainly affect overall reaction speed, if no then probably not.
[Edited on 13-3-2013 by blogfast25]
|
|
|
elementcollector1
International Hazard
    
Posts: 2684
Registered: 28-12-2011
Location: The Known Universe
Member Is Offline
Mood: Molten
|
|
You know, I wonder what it would do to put a piece of sodium in a glass tube or sep funnel at the top of the condenser, to see if it really
is atmospheric H2O or O2 that's destroying my K. I have plenty of sodium. This probably won't be my production method, but it would be another
interesting test (also it would be interesting to see what the sodium metal does). Now, how can I make some sodium wire? Would a plastic pipette be
strong enough, or would the sodium need to be softened first?
Elements Collected:52/87
Latest Acquired: Cl
Next in Line: Nd
|
|
|
violet sin
International Hazard
    
Posts: 1480
Registered: 2-9-2012
Location: Daydreaming of uraninite...
Member Is Offline
Mood: Good
|
|
http://youtu.be/btvQdW4fXJE
youtube vid. the guy uses a clay extruder from an arts and crafts supply store to make Na wire. worth a quick look.
|
|
|
condennnsa
Hazard to Others
  
Posts: 217
Registered: 20-4-2010
Location: Romania
Member Is Offline
Mood: No Mood
|
|
antiswatt, I think that the guy you mentioned is this guy:
http://www.youtube.com/watch?v=SCiP-COawoU
http://www.youtube.com/watch?v=0pz0qB9yKVk
there is also a patent from 1894 claiming that it's possible to melt kno3 in an aluminum container and electrolyze it to potassium and NO2
here it is
https://docs.google.com/a/google.com/viewer?url=www.google.c...
|
|
|
| Pages:
1
..
47
48
49
50
51
..
66 |