| Pages:
1
2
3
4 |
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
Yes, indeed. I have both urea and sulfourea, and happen to be running an experiment since Wednesday on both of them. xUrea+Formic acid (1 drop), in
an ester.
I haven't gotten metal plating from a solution containing either of them; but they are quite conductive.
Urea also works well for increasing solubility of various salts both in molten form, and not.
My experince so far: Urea has a low electrochemical resistance window, and tends to be a little too easily broken down into hydrogen, CO2, and
ammonia. Sulfourea is less reactive. I think it may be more reducing.
But that causes precipitation of sludge in a lot of experiments.
I tried urea + CholineChloride a few months ago at ?Draconic Acids? request; It ended up turning black over-night due to electrolysis and plated
sludge, only. I'm not sure how to tame it ... 
|
|
|
DraconicAcid
International Hazard
    
Posts: 4412
Registered: 1-2-2013
Location: The tiniest college campus ever....
Member Is Offline
Mood: Semi-victorious.
|
|
Well, it was an idea.
Please remember: "Filtrate" is not a verb.
Write up your lab reports the way your instructor wants them, not the way your ex-instructor wants them.
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
Idea are welcome. I am an experimentalist. I like to test out ideas. 
|
|
|
bnull
National Hazard
   
Posts: 596
Registered: 15-1-2024
Location: Home
Member Is Offline
Mood: Sneezing like there's no tomorrow. Stupid cat allergy.
|
|
I came across a 2009 patent about mixtures of nitrates for use in heat transfer. You may be interested to try one of them, if you haven't yet. I have
no clue about the solubility of nickel salts in these eutectic mixtures.
Attachment: US7588694 - Low-melting nitrates for heat transfer.pdf (957kB)
This file has been downloaded 130 times
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
I have potassium nitrate and ammonium nitrate on hand. I've tried molten mixtures of these; I also have sodium nitrite.
I don't have lithium nitrate or calcium nitrate on hand.
I do have calcium hydroxide, and calcium carbonate, sodium carbonate, and lithium carbonate; so I might be able to use ammonium nitrate + alkali
carbonate, to generate the missing salts. But as always, my ability to predict outcomes of chemical reactions is pretty weak. So, I'm not sure how
to check if the proper products have been created.
Looking at the paper, the first thing that comes to my attention is these are hydrated salts (quad-hydrate.)
And removal of water is quite difficult where nitrates are concerned. Nitrates are very prone to giving up oxygen at the anode, as gas, and
decomposing.
I've found that nitrite salts, and sulfite salts; are a little more stable in electrolytic cells.
Sodium Nitrite, Sodium sulfite, ( but not sodium metabisulfite), can be made to conduct for quite a while without rapid decomposition.
Is there any particular reason you found this paper interesting; or is it just the fact that it's molten at very low temperatures?
Alum (K-Al sulfate, hydrates), are extremely fusable at low temperatures as well; but again, the presence of water is a problem. They tend to
decompose. I have aluminum sulfate, anhydrous, and potassium hydroxide, and have been exploring alum production during electrochemical reactions.
This is one of the ideas I may pursue this week, with urea. (Note: All other urea experiments this week failed. Not worth reporting. )
In your copper plating solution with Urea, what source did you use to obtain the copper oxide? I have copper sulfate on hand, which is easy to
convert to copper carbonate; but I'm not sure if I hit the sulfate with a torch, if it would decompose to an oxide easily. If it had enough
conductivity to plate, it's an experiment I'd like to try replicating even though it's not nickel based. I am able to plate copper in water, very
successfully, but I've not tried it in ionic liquids yet.
If you were able to get clean copper to plate out of the solution, I'd be amazed.
|
|
|
bnull
National Hazard
   
Posts: 596
Registered: 15-1-2024
Location: Home
Member Is Offline
Mood: Sneezing like there's no tomorrow. Stupid cat allergy.
|
|
Not exactly. The only hydrated salt is calcium nitrate. The other are hygroscopic but not hydrated.
During college, while I was browsing the Physics Library, I found a method to generate small quantities of sodium from a mixture of sodium nitrate and
nitrite using a small lamp as electrode/vial. It came from a 1960s book series on experimental physics, of which I copied the section to a sheet that
is buried somewhere in my papers. Since I can't find it now (it's about eight years of loose sheets in no specific order, stored everywhere) or visit
the library, I searched and found the original paper: a thesis by Robert C. Burt, "Sodium by Electrolysis through Glass" (attached). That's what
prompted me to suggest the molten mixed nitrates. Supposing nickel is soluble in the mixture and is plated out of the solution at lower voltages than
the alkaline metals, it seemed feasible to me. The lower temperatures made it even more interesting.
Quote: Originally posted by semiconductive  | | In your copper plating solution with Urea, what source did you use to obtain the copper oxide? I have copper sulfate on hand, which is easy to
convert to copper carbonate; but I'm not sure if I hit the sulfate with a torch, if it would decompose to an oxide easily. |
Copper sulfate plus sodium carbonate. The usual way: decant, filtrate, wash several times with water, dry, heat while stirring until it becomes black.
There's no secret. I guess you don't even need to convert it to oxide but, well, I had the oxide and no carbonate.
Quote: Originally posted by semiconductive  | If it had enough conductivity to plate, it's an experiment I'd like to try replicating even though it's not nickel based. I am able to plate copper
in water, very successfully, but I've not tried it in ionic liquids yet.
If you were able to get clean copper to plate out of the solution, I'd be amazed.
|
I only tested the conductivity. It plated but the copper was easily removable because the conditions were unfavorable (me turning the dial from side
to side to see where the solution began conducting, and checking what happened to the copper surface on each electrode: one shiny, the other covered
in new copper).
Attachment: R. C. Burt - Sodium by Electrolysis through Glass.pdf (2MB)
This file has been downloaded 158 times
Edit: Typos again.
[Edited on 1-7-2024 by bnull]
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
OK. I read the paper(s). Yes, I see now that only the Ca salt was a hydrate; the way the first sentence was written was unclear to me.
They also are able to successfully dehydrate (or mostly) by heating to 150C. This could easily be done with my electric mantle.
In the second paper, R.C. Burt, he notes that the molten salt releases NO3 gas during electrolysis, and that's perfectly consistent with my
observation of alkalai-NO3's willing-ness to decompose during electrolysis.
The issues I suspect, then, will be whether or not the NO3 will prefer to gassify rather than to dissolve nickel anodes; and secondly, what the
presence of nickel does to the Eutectic mixture's melting point.
These salts are mildly strong oxidizers, so I'll have to see what they do with kerosene and silicone oil. I will probably dessicate them first,
exposed to air; at about 150C; but I can probably get away with them under kerosene or silicone oil if melting carefully at <95C.
One other question comes to my mind, and that's how much carbonate presence will cause precipitation/hardening/melt temperature change.
I only have Lithium in chloride and carbonate forms right now.
I'll start by replicating your urea and copper oxide formation idea; I'm going to mix sodium hydrogen carbonate, with copper sulfate, and I'll attempt
to to avoid making the resulting precipitate too alkalai; because that might re-dissolve the copper carbonate. At least, that's what I recall being a
source of failure the last time I tried it. Then I'll torch it, to see how easy it is to oxidize.
260mg CuSO4 pentahydrate, 87mg NaHCO3, should have enough carbonate to totally make CuCO3; but it will likely be basic Cu2OH2Co3, or something like
that. I'll add in steps, and stop if the water goes clear early.
But: I only have half the sodium, to make sodium sulfate (Na2SO4); therefore it's possible that the reaction won't go to completion or will be slow;
Na-H-Co3 + Cu SO4 -?-> Na-H-SO4 + CuCo3
I dissolved 260mg CuSO4 in 50mL reverse osmosis water; observed some bubbles sticking to glass, which is typical since RO water has some CO2
pre-dissolved.
I Dissolved 87 [mg] of baking soda, in 5 ml of RO water. Same bubble formation issue observed.
I waited untill CuSO4 completely dissolved, and then added 2.5CC of the NaHCO3 solution to it.
Coloidal suspension immediately forms in the beaker. No CO2 gas evolution observed. Cover with cap to keep bugs out, and let it sit. Will check it
later.
Edit: After 2 hours, precipitate has settled. Water is still very slightly blue, so I went ahead and added the remaining sodium bicarbonate. It's
clear enough, though, that I think the majority of the copper has already been removed. I may not get a complete reaction yield, this way; but I
think it good enough. I'll just remove water by pippette, and rinse it three times over the next day or so.
Rinsing complete; 3x 100 mL -- I have a nice even precipitate layer.
It has the azurite/malachite blue green color spectrum, which I expected.
[Edited on 2-7-2024 by semiconductive]
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
The precipitation gave 72 [ mg ] of basic carbonate from 260 [mg] of pentahydrate.
I have 1.04₂ [ m · mol ] of copper
Cu=63.546 [ g / mol ] CO₃=60.01 [ g / mol ] OH- = 17.008 [ g / mol ]
Cu₂ · (OH)₂ · CO₃ = 221.₁ [ g / mol ]
Assuming perfect conversion to basic carbonate, I ought to have: 115 [mg] of product.
72/115 ≈ 60% conversion.
Hmm ... I'll have to try this again, later, with double the sodium carbonate and see if the conversion efficiency goes up. I got more than half
converted, so it seems at least some Sodium Hydrogen sulfate was formed.
Test tube oxidizes nicely to copper oxide. Yes!
About how much urea, do you think, would be appropriate for a first attempt?
|
|
|
bnull
National Hazard
   
Posts: 596
Registered: 15-1-2024
Location: Home
Member Is Offline
Mood: Sneezing like there's no tomorrow. Stupid cat allergy.
|
|
Quote: Originally posted by semiconductive  | | Hmm ... I'll have to try this again, later, with double the sodium carbonate and see if the conversion efficiency goes up. I got more than half
converted, so it seems at least some Sodium Hydrogen sulfate was formed. |
I always used an excess of sodium carbonate. Copper carbonate (I should call it basic copper carbonate but old habits die hard; sorry for that) is not
much soluble in sodium carbonate as it is in sodium hydroxide. At least that's what I had observed long ago.
Hmm... From 500 mg to 1 g of urea. I don't know the solubility of copper oxide in urea; I suppose that it dissolves as an amino complex, given the
blue color and the smell of ammonia after dissolution. The color when molten is close to this deep
blue. Melt urea, add a bit of copper oxide, shake the tube, insert electrodes, and add more copper oxide as the color weakens.
[Edited on 4-7-2024 by bnull]
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
Hmm.
The copper reduced to a fine oxide powder in the test tube; kind of greenish-grey/black. ( Slightly less than 400C cooking temperature, roughly 35
watt ).
It's a bit of a pain to work with this small amount of copper oxide (I'm mildly disabled. )
So, I'm going to just put about 1/2cc worth of urea prills on top of it, cover with kerosene (which just acts as an air barrier), and melt the urea
into the powder. This ought to make bubbling from 'fizzing' as you called it, visible.
I can make more copper oxide, later, if this shows any promise at all.
Picture of test tube just before capping with kerosene.

|
|
|
bnull
National Hazard
   
Posts: 596
Registered: 15-1-2024
Location: Home
Member Is Offline
Mood: Sneezing like there's no tomorrow. Stupid cat allergy.
|
|
Quote: Originally posted by semiconductive  | | So, I'm going to just put about 1/2cc worth of urea prills on top of it, cover with kerosene (which just acts as an air barrier), and melt the urea
into the powder. This ought to make bubbling from 'fizzing' as you called it, visible. |
It will be very visible: it will foam a lot. I did today as you said, putting urea on top of copper oxide but without kerosene. I don't know if it
would stop the foam from rising up the test tube (probably, yes). When solidified, it looks like the picture below.
A couple of pictures of the copper complex (tetraammine, or so it seems) dissolved in molten urea from yesterday.
 
Edit: Typo.
[Edited on 7-7-2024 by bnull]
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
I have a lot of things to report. I took a bunch of pictures, and can post images for whatever is of interest; but I'd rather not post 100 shots of
nothing... 
I put kerosene on top; and heated the kerosene rather than the urea. This caused a slow melting process of the urea and gave me good control. There
was significant gas coming off the urea as it turned blue. It did not foam, at all, in my experiment.
It just released gas, some of which I think was steam.

Conductivity rose before the urea had melted. I used a copper anode and a graphite cathode. It rapidly rose in conductivity as it started to melt,
in fact it was too conductive. I had to put a resistor limiter in to reduce the electric heating. I've already got a 30W soldering iron touching the
test tube from behind (the philips screw on it is just visible in the picture. ) This is enough heat.
Overcurrent, like I had, creates side products. I'm not sure how much it might have affected the rest of the experiment.
It's pretty obvious that the copper is extremely reactive with urea. And from the odor, I think copper oxide encourages transformation of urea into
ammonia when water is present. The slight ammonia odor went away as the reaction progressed.
For my next attempt, I won't bother to oxidize the copper; because I'd like to test if urea is reactive enough to decompose carbonate; and if the
ammonia smell will be avoided.
A couple of times, I got it too hot; but quickly reduced temperature. If significant gas came off the anode, I would cool it.
As the urea finally melted, I could see a thin layer of copper form on the graphite electrode. It re-dissoved shortly thereafter. The color reminds
me of ammonia-copper etching color, sort of off-color salmon pink/brown. Once the urea was finally melted, I removed the graphite electrode; cleaned
it, and replaced it in the liquid to make sure there was nothing on the electrode interfering with plating.
No furthur plating after returning it to service for two hours. The current level was easily adjustable from 3mA to 45mA, and gas in proportion to
the current was forming at the electrode.
The color was so dark blue that it was difficult to see the electrodes. So I added about another 1/2 CC of urea prills on top. I watched the blue
liquid wick up onto them as they melted. It's quite obvious that the melting point of the blue liquid was *much* lower than that of the urea prills.
And it's also obvious that adding extra urea was raising the melting point of the whole mixture (not desirable.)
The liquid slowly darkened again, and I thought I had added too much heat. Since the tube was messy, I decided to change the kerosine and the test
tube. After letting it solidify, I decanted; broke up the solid urea and transferred it to a clean test tube. I could see the solid chunks were
slightly greenish in color, except for where the air could reach it, where it became pure blue again.
This makes me suspect that the blue color is actually a hydrated ammonia ion / ligand. But: Under kerosene, (hypothesis), the mass turns green slowly
as it drys; and it's melting termperature rises, cauing solidification.
After quite a while, it would no longer melt. I raised the heat, and am not sure if I caused decomposition or not. But, it still conducts
electricity even when semi-solid. It is not until the temperature becomes almost room temperature that conductivity stops.
At this point, I decided to add an ester that is liquid. I was hoping it would lower the melting point of the mixture. It did; although most of the
mass remained solid. However, after about an hour and a half I could see the ester starting to brown; which means the heat was well above the melting
point of normal urea.
On the other hand, in the presence of ester; copper began plating on the electrode.
It's dingy, not bright; but it still was plating out; This is at 2 [mA] current.
Shall we try again?
Suggestions / questions ?


[Edited on 7-7-2024 by semiconductive]
|
|
|
bnull
National Hazard
   
Posts: 596
Registered: 15-1-2024
Location: Home
Member Is Offline
Mood: Sneezing like there's no tomorrow. Stupid cat allergy.
|
|
Thank you for your observations. I didn't expect that much from urea. Amazing.
Quote: Originally posted by semiconductive  | | I put kerosene on top; and heated the kerosene rather than the urea. This caused a slow melting process of the urea and gave me good control. There
was significant gas coming off the urea as it turned blue. It did not foam, at all, in my experiment. It just released gas, some of which I think was
steam. |
And possibly a lot of ammonia.
Quote: Originally posted by semiconductive  | | It's pretty obvious that the copper is extremely reactive with urea. And from the odor, I think copper oxide encourages transformation of urea into
ammonia when water is present. The slight ammonia odor went away as the reaction progressed. |
Yes,
apparently. CuO hydrates to copper hydroxide and then decomposes urea to ammonia and carbon dioxide, which could explain both the effervescence and
the blue color. Edit: Most probably there's no hydration and copper oxide in the presence of water catalyses the decomposition of
urea, liberating ammonia and carbon dioxide (if so, then [CuO+H2O] behaves as a strong base; how odd). Like
this:H2O(l)+(NH2)2CO(l)CuO/Δ→2NH3(g)+CO2(g).
Quote: Originally posted by semiconductive  | | As the urea finally melted, I could see a thin layer of copper form on the graphite electrode. It re-dissoved shortly thereafter. The color reminds
me of ammonia-copper etching color, sort of off-color salmon pink/brown. |
Strange. It reminds me of an
oscillating reaction with iron electrodes in acid; it involved passivation and depassivation. Schönbein had discussed it with Faraday for a while. In
the present case, it could be a critically damped oscillation.
Quote: Originally posted by semiconductive  | I could see the solid chunks were slightly greenish in color, except for where the air could reach it, where it became pure blue again.
This makes me suspect that the blue color is actually a hydrated ammonia cation. Under kerosene, the mass turns green slowly; and it solidifies.
|
The green color may be due to the formation of a copper(ii)-urea complex (see, for example, Omar B. Ibrahim,
Complexes of urea with Mn(II), Fe(III), Co(II), and Cu(II) metal ions).
Quote: Originally posted by semiconductive  | After quite a while, it would no longer melt. I raised the heat, and am not sure if I caused decomposition or not. But, it still conducts
electricity even when semi-solid. It is not until the temperature becomes almost room temperature that conductivity stops.
At this point, I decided to add an ester that is liquid. I was hoping it would lower the melting point of the mixture. It did; although most of the
mass remained solid. However, after about an hour and a half I could see the ester starting to brown; which means the heat was well above the melting
point of normal urea.
On the other hand, in the presence of ester; copper began plating on the electrode.
It's dingy, not bright; but it still was plating out; This is at 2 [mA] current. |
Do you think it possible
that the ester you added generated a copper salt that is soluble in urea and from which copper plates out more easily, while the organic anion reacts
with the copper complexes in solution, repeating the process?
If you don't mind, by all means do so. It has been more interesting than I had initially
suspected.
I've been thinking of trying copper(ii) acetate. I happen to be waiting for it to
crystallise from a solution. The idea was to make large crystals but I can do that another day. If copper acetate dissolves in urea without
decomposing it, it would be a better choice. Easier to make and purify, no need to roast it like coffee. The process would be essentially a Kolbe
electrolysis with urea as solvent. There are some questions which I can't answer for now: (1) Kolbe electrolysis of acetate works by way of free
methyl radicals. Will urea be methylated? (2) If urea is methylated, what will happen to the solution, and will copper be plated onto the electrode?
Edit: I forgot that Kolbe electrolysis is usually done with alkaline cations. I don't think it proceeds with copper as cation, hence
no methylation of urea and both questions are answered. Oh, well.
Only one suggestion. Try paraffin wax in place of kerosene. Apart from being non volatile and less inflammable than kerosene, you can pre-mix copper
oxide with it and the reaction rate with urea in the molten state slows down considerably. It will need some stirring/shaking, that's the downside.
Attachment: complexes-of-urea-with-mnii-feiii-coii-and-cuii-metal-ions.pdf (541kB)
This file has been downloaded 223 times
P.S.: Sorry for the long post.
[Edited on 8-7-2024 by bnull]
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
| Quote: | | P.S.: Sorry for the long post. |
Don't apologize! I write books unintentionally for posts, darn it!

| Quote: |
Do you think it possible that the ester you added generated a copper salt that is soluble in urea and from which copper plates out more easily, while
the organic anion reacts with the copper complexes in solution, repeating the process?
|
In the presence of water, I have observed esters breaking down into their constituents whenever acid or strong base is present. But, I didn't add
any ester until after the solution had become nearly solid and green under kerosene.
On the other hand, I think there are too many steps and possible side reaction variables for me to really guess whether it could or could not. I'll
need to do additional tests to figure that out.
The ester I used this time was citrate based, because citric acid is resistant to oxidation. I don't have enough experience to know for sure whether
it would react since this is my first attempt at copper; BUT: It generally *doesn't* react with nickel ions present.
I don't know how the ethanol I esterified it with could get displaced when the urea has already been cooking long enough to drive out any free water
and is solidifying even at high tempertature ?
I didn't see bubbling when adding the ester; so I don't think significant ethanol was released as it was hot enough to boil. eg: Adding an ester did
not make a visible reaction before electrodes were re-inserted. The picutres are bubble free.
If it did react with electrodes, the obvious product would be copper citrate dissolved in molten urea. I could make copper citrate either in water,
or perhaps (to avoid water) in methanol, DMSO, etc. ( Or suggest something simple! ) I have the citric acid; would that answer the question?
The article you linked was interesting. I have a working visible Jaz spectrometer, which could characterize the green or blue color spectrum; but
nothing that can take and compare against the FTIR spectra listed in the document. I have very little way of figuring out what kind of ligand I have
in solution, and my chemistry experience from is from college is 35+ years ago, and was my worst subject. I'm a honors BSEE, but not even an undergrad
chemist, here. 
I'm pretty sure, though, that Molten urea won't be fully spectrum tested by me; the square optical vials I have are disposable plastic and would melt.
I could take surface reflectance spectra of molten urea with a surface reflectometer in a glass test-tube, but it would have to be manually done
since the drivers for Jaz spectrometers are written in java and crash on linux systems.  . I'd love to be able to monitor ligand concentration by color, but I'm limited to manual spectroscopy at the moment unless I can find
an open source Linux driver that *works* with ocean insight spectrometers. Their support is not very good. . I'd love to be able to monitor ligand concentration by color, but I'm limited to manual spectroscopy at the moment unless I can find
an open source Linux driver that *works* with ocean insight spectrometers. Their support is not very good.
Wax; yes, I have that and occasionally use it. But it's a pain to clean out of test tubes. I have silicone oil, as well, which is less flammable; but
I've never had a flame problem without chlorates or oxide-nitrates involved; so I'm not too concerned yet. The quantity of kerosene I'm using is so
small, (Those pictures are through a microscope lens) even if it goes up it just is like a match striking.
But: I generally see smoke gathering in the tube slowly before ignition; and then there's usually not enough oxygen because of the smoke filling the
test tube.
Squirrel knocked over the CuCarbonate I set outside today; will have to re-precipitate a new batch.
Look for post below ... eventually.
|
|
|
bnull
National Hazard
   
Posts: 596
Registered: 15-1-2024
Location: Home
Member Is Offline
Mood: Sneezing like there's no tomorrow. Stupid cat allergy.
|
|
| Quote: | | If it did react with electrodes, the obvious product would be copper citrate dissolved in molten urea. I could make copper citrate either in water,
or perhaps (to avoid water) in methanol, DMSO, etc. ( Or suggest something simple! ) I have the citric acid; would that answer the question?
|
Maybe. I forgot that esters are soluble in urea; according to Clark, "[h]ydroxy compounds, ketones, esters, anilides, aminoacids, substituted
hydrazines, etc., are readily soluble and decomposition is rare." Ester would partition between kerosene and the still liquid urea. Decomposition is
rare when there is only urea and the ester in solution. He says nothing about what happens when there is more than one solute.
Still assuming that [CuO+H2O] (or Cu(OH)2, for that matter) is a strong base, and I'm skating on thin ice here, the ester would
decompose and form copper citrate, which is soluble in urea etc. If decomposition is slow, ethanol would diffuse from urea to kerosene and then
evaporate quietly.
About the linux driver, no luck here. It seems they don't give a damn about linux.
| Quote: | | Squirrel knocked over the CuCarbonate I set outside today; will have to re-precipitate a new batch. |
I know how it is, I have cats.
By the way, copper acetate is soluble in urea. There is decomposition without bubbles. It smells of ammonia and acetic acid. Again, blue color of
tetraammine complex. Copper was plating out, "dingy, not bright; but it still was plating out".
|
|
|
EF2000
Hazard to Others
  
Posts: 154
Registered: 10-5-2023
Location: The Steppes
Member Is Offline
Mood: Taste testing the Tonka fuel
|
|
Quote: Originally posted by semiconductive  | I'd love to be able to monitor ligand concentration by color, but I'm limited to manual spectroscopy at the moment unless I can find an open source
Linux driver that *works* with ocean insight spectrometers. Their support is not very good.
|
I don't know will it work or not, but there's SeaBreeze, described as
| Quote: |
device driver library that provides an interface to select Ocean Optics spectrometers. It is written in C/C++ and builds and runs on Windows (XP/7/8),
MacOSX, and Linux (x86/x64/ARM) |
Found on sourceforge: https://sourceforge.net/projects/seabreeze/, but not found anymore on Ocean Insight website.
There's also python modules based on SeaBreeze (python-seabreeze, spectrabuster), but they are more for automating the process. Almost anyone would prefer a GUI (I apologize to any robots reading this).
Wroom wroom
"The practice of pouring yourself alcohol from a rocket fuel tank is to be strongly condemned encouraged"
-R-1 User's Guide
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
Thanks.
| Quote: | | I don't know will it work or not, but there's SeaBreeze, described as... |
Downloaded, transferred to my Raspberry PI™ (ARM platform) in the lab.
From the documents, this is restricted to the USB cable and not TCP/IP.
Still, that's fine. IF it works at *all* that's better than what I've got now.
| Code: |
cd seabreeze-3.0.11/SeaBreeze
make
|
and .. no plug and play joy.
| Quote: | ../../../include/common/features/Feature.h:62:13: error: dynamic exception specifications are deprecated in C++11 [-Werror=deprecated]
62 | throw (FeatureException) = 0;
|
This is a warning elevated to an error; which means seabreeze is basically not being maintained and has grown old;
Edit: The MIT license for SeaBreeze is 2014; so this code was using depreciated coding on the day it was written.
| Quote: | | Almost anyone would prefer a GUI |

I prefer something that actually works on a Raspberry PI™ which is the educational platform, open source, (de-facto) used throughout the world; and
using it without charging me industrial prices for a hobby.
I don't mind writing a python script to control soldering iron temperature in response to SeaBreeze feedback.
I'll attempt to use a USB RS232 dongle, to turn on and off a 110VAC outlet so I can temperature regulate the test tube via soldering iron and monitor
the color change vs. time.
[Edited on 9-7-2024 by semiconductive]
This bug has been known since 2017, fix is to set compiler to --std=c++03
https://sourceforge.net/p/seabreeze/tickets/34/
But still no joy, new error:
| Quote: | PixelBinningFeatureAdapter.cpp:58:14: error: catching polymorphic type ‘class seabreeze::FeatureException’ by value [-Werror=catch-value=]
58 | } catch (FeatureException) {
|
I will try:
Editing file, src/api/seabreezeapi/PixelBinningFeatureAdapter.cpp
I Searched for every catch statement, and put an ampersand after the constant being caught:
line 58, for example:
| Code: | } catch (FeatureException&) |
Still no joy, new error in a different file.
| Quote: | | NativeUSBLinux.c:35:10: fatal error: usb.h: No such file or directory |
What?! usb exists on a raspberry PI!
... thinking ...
| Code: |
# find / -iname usb.h
# ... /os-support/windows/WinDDK_Includes/usb.h
# ... /include/native/usb/USB.h
|
Hmm .. but I don't have a system header in lowercase, called usb.h , on the raspberry PI -3 ™.
Annoying. I'm going to have to upgrade all the software on the Raspberry PI ™ just to see what the header file name is for the USB system the PI
already has.
| Code: |
$sudo apt-get --fix-missing upgrade
$sudo apt-get update
# Rebooted here, didn't start correctly, but had white screen with waste basket.
# I logged in using ssh, and reconfigured apt-get:
$sudo apt-get --configure -a
# Now, to re-install the usb package; I need to know what version of library is used:
$ldconfig -p | grep libusb
libusb-1.0.so.0
# The header file is always in the development version, so install dev version:
$sudo apt-get install libusb-1.0.0-dev
$sudo apt autoremove # Clean up any unused packages.
|
re-running make now complains about a missing USB.h, that is *upper* case; from the same line! 
I know that file exists in SeaBreeze as /include/native/USB.h
What educated i***t made gcc report an upper case file as lower case, before...
Oh well, I've shown how to update Rasperry pi ™ as a bonus.
Editing, src/native/usb/linux/NativeUSBLinux.c
commenting out line 35, since <USB.h> is a c++ header, and this is a *C* file.
And now I have a bunch of undefined linux functions. Replacing line 35 with: #include <llibusb-1.0/libusb.h> // fixes nothing.
Checking kernel headers, and Linux USB native does not have the missing functions .
Checking the SeaBreeze readme.txt, I see it! They want libusb version 0.1 for Linux.
I'm not doing that. I'd end up breaking my raspberry pi which uses version 1.0 already.
so, time to manually upgrade seabreeze to usblib-1.0.
Small headache! I've got to read a bunch of api names, and changes.
More to come *if* I'm able to debug SeaBreeze device driver....
[Edited on 10-7-2024 by semiconductive]
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
I successfully ported SeaBreeze's native linux USB to use a modern libusb-1.0
SeaBreeze's USB interface will now will compile on any Raspberry PI.
I see the jazUSB being built, which is my spectrometer.
Therfore, I'm getting excited  This might actually work, and not be a waste
of time. This might actually work, and not be a waste
of time.
The package is almost completely built, but ... 
In file src/common/Log.cpp, false indentation had to be deleted in two places.
And after that, I got this weird error which took a while to figure out:
| Quote: |
BlazeUSBTransferHelper.cpp:81:49: error: ‘void* memcpy(void*, const void*, size_t)’ writing to an object
std::vector<unsigned char>’ with no trivial copy-assignment; use copy-assignment or copy-initialization
=class-memaccess]
81 | memcpy(&outBuffer[0], &buffer[0], length);
|
It'm not sure it's really a bug; and I can forced the file to compile by explicitly typecasting the buffers to (void*).
Example:
| Code: |
memcpy( (void*)&outBuffer[0], (void*)&buffer[0], length )
|
On to different bugs ... or rather, repeats of earlier bugs in new places:
Polymorphic catch errors show up in the file:
src/vendors/OceanOptics/features/data_buffer/DataBufferFeatureBase.cpp
It's an easy fix: search for every catch statement, look for word fpnfe in it, and prepend an ampersand:
eg:
| Code: | catch (FeatureProtocolNotFoundException &fpnfe) |
Same kind of bug in file: src/vendors/OceanOptics/features/light_source/LightSourceFeatureBase.cpp
There is a "catch( FeatureProtocolNotFoundException ex )", that needs an & before the "ex".
And 4+ more of them in file: /src/vendors/OceanOptics/features/spectrum_processing/SpectrumProcessingFeature.cpp
And 4+ more of them in file: src/vendors/OceanOptics/features/thermoelectric/ThermoElectricFeatureBase.cpp
And 6 more of them in file: src/vendors/OceanOptics/features/pixel_binning/STSPixelBinningFeature.cpp
At this point, a handful of "notes" scrolled by which I'm going to ignore for now.
And the make exited all directories and tried the final link of 'test', at which point it bombs because the library it needs to link is not -lusb
I edit "common.mk", and find the linux part and the flag "-lusb" is, and just change it to "-lusb-1.0"
The SeaBreeze driver now compiles against libusb-1.0. YAY !!!!!!
There is an annoyance in: sample-code/c/demo-pthreads.c
The 'snprintf' commands, need to be replaced by 'sncat' commands.
on line 164
| Code: |
strncat( line, msg, sizeof(line)-strlen(line)-1 );
|
and line 324
| Code: |
strncat( line, devices[i].serial, sizeof(line)-strlen(line)-1 );
|
And, finally, that's the last of the bugs.
Make finishes building SeaBreeze with no errors.
Therefore: I have all of SeaBreeze and test programs installed on my Raspberry PI ™. 
Now to actually find a USB cable, plug in my spectrometer ... and see if it works.
EDIT: Yes !!!!!!!!!! it DOES! This is AWESOME.
There is a single bug that I noted during testing; Linux does not call USB close when when signals are caught. If your seabreeze application is
killed by a unix signal; the USB interface will be left *claimed* as if open. This results in the spectrometer not being openable again, until a USB
reset or power-cycling happens.
I have a Jaz, so I reset my spectrometerer with vendorID (Ocean Optics), 0x2457, and product Jaz 0x2000.
| Code: |
sudo usbreset 2457:2000
|
I have also just found an ocean optics Raman Spectroscope within my price range which SeaBreeze has a driver for.
Hopefully, I can find or make a power supply for it; otherwise I'll have to watch for another one in the coming months.
Here's a patch file to fix SeaBreeze, in case anyone else wants to use my upgrades.
Attachment: seabreeze-3.0.11a.diff (41kB)
This file has been downloaded 106 times
[Edited on 12-7-2024 by semiconductive]
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
Copper sulfate, pentahydrate 255 [mg] in 45 ml R.O. water.
+ 170 [mg] NaHCO3 in 5 ml water.
Extra metallic support on 5 sides, makes a partial Faraday cage.
My experiment ought to be safe even if lightning strikes.
Scheduled to change water 3x rinsing precipitate thoroughly, through Saturday 7/13.

|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
There was a slight loss of material during transfer; and I forgot to cover the beaker with a watch glass.
So, this might be 1 or 2 mg in error.
The squirrels didn't invade this time, though.  Success is good. Success is good.
The precipitation gave 110 [ mg ] of basic carbonate from 255 [mg] of pentahydrate.
I have 1.02₁ [ m · mol ] of copper
Cu=63.546 [ g / mol ] CO₃=60.01 [ g / mol ] OH- = 17.008 [ g / mol ]
Cu₂ · (OH)₂ · CO₃ = 221.₁ [ g / mol ]
Assuming perfect conversion to basic carbonate, I ought to have: 112.₉ [mg] of product.
110/112.₉ ≈ 97.4₃ % conversion.
I've gotten pretty close to theoretical maximum yield.
My spectrometer is also giving me data samples, although the data is coming into my laptop raw. It doesn't give the data after the spectrometer
processed it. A bit annoying. The data also has noise spikes.
I'm writing some software to filter the data, statistically, since I don't have an operational ocean optics software package to process it. I'll get
a good color scan of the basic copper carbonate, and post it (eventually) below. I'm slow, but I am getting there!
I've also ordered some bluetooth outlets which will allow me to robotically control various apparatus during experiments and do closed loop
temperature control.
And I bought two Owon BT41 Digital multimeters, that can monotor temperature and current, during the experiment; but just realized I should have
bought a third one to monitor voltage. Oh, well, for this second experiment we'll just have a current profile and temperature record.
I tested my DMM's out today, and am able to control them perfectly and record data on my Rasperry Pi.
So, I ought to be able to build a precision automated test bench, relatively soon.
The seller of the Raman spectrometer head hasn't responded to my inquiries. So, I don't think I will risk buying it. Boy, I'd love to have that,
though!
|
|
|
bnull
National Hazard
   
Posts: 596
Registered: 15-1-2024
Location: Home
Member Is Offline
Mood: Sneezing like there's no tomorrow. Stupid cat allergy.
|
|
Quote: Originally posted by semiconductive  | | I'm writing some software to filter the data, statistically, since I don't have an operational ocean optics software package to process it.
|
Did you try Spectragryph? I used it in 2019 to process flame and LED spectra I had obtained in an experimental physics lab (I still miss experimental
physics).
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
Spectragryph requires a full wine installation for linux.
It's really windows only software, closed source, that can be run under an emulator.
But, it does look cool. I'm just interested in getting something to work, open source, if possible.
eg: I dislike getting dependent on closed source software, only to have it stop working on a new release of Linux.
Python seabreeze and spectrabuster, which were listed two posts ago also failed to work.
Python seabreeze installs a binary version of seabreeze that requires libusb 0.1, which isn't installed on a Raspberry PI.
Sigh: It's too dumb to allow a new version of seabreeze (Eg: my correctly compiled version), to be used.
When I tried running the python USB version that supposedly doesn't use seabreeze, it crashed.
I've almost got a workable solution. I just need to figure out what statistical model handles the noise, best. Online literature assumes Gaussian
for data and noise, but that's lazy crap and my tests show it's grossly inaccurate. I'm going to try a uniform distribution combined with a binomial
distribution and see if I can identify outlier samples with it, reliably. Should only take a few days to guess a decent model; then I can use the
tungsten light source's blackbody radiation to create a highly accurate calibration curve for the sensor. The code to do all of this is probably only
one page of Python text; which can run on *any* platform Windows ™, Mac™, or Linux. It's worth doing for open education/amateur purposes.
I've just got to figure out a general formula for adding variances of dis-silimar distributions, with different number of samples.
I'm close, as I've already been working on a similar problem:
https://physicsdiscussionforum.org/probabiility-and-statisti...
[Edited on 17-7-2024 by semiconductive]
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
I'm winning the battle. I got a background noise fingerprint.
My Jaz spectrometer sensor looks like this in the dark, with thermal drift happening over 8 hours.
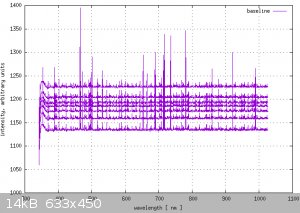
Each line is 4096 samples, at 1 second apart; so roughly an hour's worth of data.
The noise is fairly repeatable, like a fingerprint, but I see a few of the sensor noise peak's change visibly in the plot ... which is not good.
I'll probably let this run for another 24 hours, use the data to see if any of my pixels are problems; Then see about writing a filter program to
reduce errors by injecting anti-correlated noise.
After that, I just need to calibrate the intensity scale against the built in Tungsten lamp. eg: It's time to look up blackbody radiation curves ...
and refresh my memory. Power per frequency, not power frequency is what I recall the formula being; so I need to figure out how to convert power
density into total energy at a wavelength.
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
I bought some TPLink™ KASA™ plugs, since those are on the market and can be controlled by a Raspberry PI™ using python; therefore qualified
people with safety equipment can duplicate what I've done. This is just for educational purposes, only and I'm not recommending it. The KASA plugs
use reed-relays, which means they will wear out; But, it's good enough for running a soldering iron. I'll report any failures if they occur and
lifespan.
This is a picture of urea under kerosene with the soldering iron in the background.

There is about 3cc's of kerosene in the test tube, and I have metallic shielding in case of fire as I don't trust KASA yet to run un-attended. There
is roughly 1/2cc's worth of Urea Prills in the tube with a 30W soldering iron behind it. This is wired solidly to a chemistry clamp stand.
My multimeter says the tube is at 132.1 [ °C ], with the KASA program pulse width modulating the iron at 72% of full power via 1.5 second long
pulses. The tube's been heating all afternoon and stays within ±1 [ °C ] stably.
Notice: no prills are melted, yet. I expect the Urea is now quite dry, with excess moisture driven off.
At room temperature, my ohm meter's thermal probe registered 3 degrees celsius higher than the wall thermostat does. So, it's possible the
temperatures I report might need adjusting downward by up to 3 [ °C ]. I haven't calibrated the probe more precisely than that.
The wall barometer, shows 29.85 inches of water at my elevation. But, kerosine at these temperatures is not very volatile and will remain in the test
tube for several days without issue.
A quick web search shows the lowest listed melting point of Urea as, 132.7 [ °C ]
I don't know how accurate that is.
https://www.accessscience.com/content/article/a722900
I will continue to raise the temperature this afternoon at 1 degree C every two hours, and report what temperature the urea prills start to melt at.
I'll be checking for decomposition smells, and stability of the liquid also.
No odors, yet, except for slight oily feel of warm kerosene.
The following program is what I'm using; and if it gets interrupted during an on cycle -- it can cause problems by leaving the switch permanently on.
Fix it, or use at your own risk.
https://pypi.org/project/python-kasa/#description
| Code: |
#!/bin/env python
# Script to run KASA heating plug
# This is a gnu public license, 3.0, hack.
# Andrew Robinson of Scappoose, July 2024.
# https://www.gnu.org/licenses/gpl-3.0.en.html
import time
import asyncio
from kasa import Discover
async def main():
count=0
devices=await Discover.discover()
for dev in devices.values():
await dev.update()
power, maxpower = 0.50, 0.80
while (dev.alias == "heat"):
# Power increase rate:
# .00015 roughly one percent per hour at 50%
# .0015 roughly ten percent per hour at 50%
# .0045 roughly twenty five percent per hour hour at 50%
# Rates will slow at higher powers, purposely
# this allow rapid warm ups but slower sweeps of hot temperatures.
power+=.00001
if (power>maxpower):
power=maxpower
s=1.5+20*power**3
print("\r Heat Power %8.5f "%power,end="")
await dev.turn_on()
await dev.update()
time.sleep( s )
if (power<1.0):
await dev.turn_off()
await dev.update()
time.sleep( s*(1/power-1) )
if __name__ == "__main__":
asyncio.run( main() )
|
[Edited on 26-7-2024 by semiconductive]
Good morning.
I'm not sure what to say ... the repeat experiment did NOT go as planned.
The Urea, unadulterated, did not melt under Kerosene.
The prills are still quite visible. Although the Urea does appear to have partially dissolved into the Kerosene; and re-crystallized after cooling.

I've ordered two more Owon BT41 meters, to double check the thermocouples.
It sort of looks like a design flaw in the meter; but since this is one of the few meters that actually works with Linux and windows, both, over
bluetooth,
I'd really like to be able to use it with my Raspberry PI.
I also notice that it registers 0 degrees celsius about once in 100 readings; so the meter's sampling algorithm is flawed, too. ( Something I can
write software to detect and reject, though. )
I've got a difference between the two meters that I have of about 6 degrees celsius. It's consistent. They're really inaccurate. I'm not sure what
the temperature actually were, though, during the experiment. Argh!
I had an enclosed K type thermocouple in a steel sleeve for a different meter, and plugged it into the Owon only to get readings which were much
worse.
I'm dipping one of the thermocouples into silicone, to see if I can at least make a temporary shield for it. But, this is not a really good idea,
because silicone is chemically reactive.
I need to either figure out how to make a teflon sleeve, or get my inert gas setup finished so I can make a true glass sleeve. Stainless steel
thermometers aren't really a good idea. Setbacks ...
[Edited on 26-7-2024 by semiconductive]
|
|
|
semiconductive
Hazard to Others
  
Posts: 326
Registered: 12-2-2017
Location: Scappoose Oregon, USA.
Member Is Offline
Mood: Explorative
|
|
Note: heating rates are way off in heating script of last post as I changed s formula after comments, but I can't edit to fix the comments any more.
It's closer to power+=.0045 makes a 25% change in 10 minutes, not in an hour.
eg: .0009 is 5% change in 10 minutes.
You'll need to experiment to figure out the other rates.
I calibrated one Owon thermometer after dipping it in orange sensor-safe silicone from permatex.℠. It reads 5 degrees C high; but this is
consistent at all temperatures so I wrote a program to adjust the temperature.
Reheating, again, using power+=.0009 # 5% in 10 minutes, 40% in an hour @ 19% start.
The thermocouple is at the front of the tube, where it should be coolest.
Most of the test tube de-fogged by 30 [°C]. But: The dust around the prills did not evaporate until about 108 [°C]. Picture is 110 [°C]. The
prills are not melting.

130 [ °C ] @ 72.9% of 30W power.
131 [ °C ] @ 73.3% ...
132 [ °C ] @ 74.0% ...
133 [ °C ] @ 74.6% ...

134 [ °C ] @ 75.0% ...

135 [ °C ] @ 75.8% ...

137 [ °C ] @ 77.3% ...

139 [ °C ] @ 78.6% ...

141 [ °C ] @ %80.0 ...

143 [ °C ] @ %81.3 ...
144 [ °C ] @ %82.0 ...
145 [ °C ] @ %83.0 ...
146 [ °C ] @ %83.8 ...
147 [ °C ] @ %85.4 ...

148 [ °C ] @ %87.2 ...
149 [ °C ] @ %88.0 ...
150 [ °C ] @ %88.75 ...

I don't get it.
This is hot. Burns my finger, hot. Not even the tops of the urea prills have melted.
Does driving off water slowly before melting raise the melting point???
151 [ °C ] @ 89.5%
152 [ °C ] @ 90.5%
153 [ °C ] @ 91.3%
154 [ °C ] @ 92.2%
155 [ °C ] @ 93.2%
I give up (for today). Stumper.
The silicone part that was submerged in hot kerosene, softened. Silicone's weakness is gasoline; and Kerosene is not far from it. But, the silicone
dip survived intact; it's good enough, I can use silicone for a while.
( AND YES, those really are different pictures taken a few minutes apart. )
[Edited on 27-7-2024 by semiconductive]
|
|
|
| Pages:
1
2
3
4 |
|