| Pages:
1
2 |
Sulaiman
International Hazard
    
Posts: 3692
Registered: 8-2-2015
Location: 3rd rock from the sun
Member Is Offline
|
|
My comments above give the impression that I favour mercury u-tubes etc.
I definitely do not,
I use and recommend Bourdon gauges of the type shown in the first few posts.
There are so many options for pressure/vacuum gauges but the cheap type are my choice.
CAUTION : Hobby Chemist, not Professional or even Amateur
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
Are vacuums (<10mmHg) expensive??
I need 28 Inches Hg or higher.
Here is my conceptualization of the vacuum distillation setup minus the gas trap (the one that doesn't implode right?)
Which leads me to my next question, what kind of a trap do I need between my vacuum and the vacuum takeoff adapter?
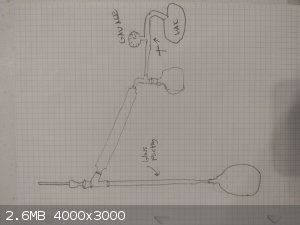
[Edited on 2/6/2023 by Yttrium2]
|
|
|
Sulaiman
International Hazard
    
Posts: 3692
Registered: 8-2-2015
Location: 3rd rock from the sun
Member Is Offline
|
|
Your diagram indicates that you want to do fractional distillations at low pressures.
I had such dreams too.
If there is a specific reduced pressure distillation that is very important to you, then go for it,
but I recommend that you work at atmospheric pressure only until you have a lot more practical experience,
by that time you will have a much better appreciation of what is required for your circumstances.
Not a criticism, just advice.
I'm still learning the hard way (mostly by failures) but I'm not in a hurry.
...........................
PS vacuum pumps for less than 10mmHg need not be expensive,
A peristaltic pump can be used for medium vacuum, low pumping rate applications
( my YT video https://m.youtube.com/watch?v=gGtYlS1ihbE)
A generic rotary pump (HVAC, car air-con service type) for medium vacuum at higher pumping rates
(either can give a gauge reading of - 1 Atmosphere)
I've not yet measured the actual minimum pressure of my rotary 
High vacuum is only for the few.
[Edited on 6-2-2023 by Sulaiman]
CAUTION : Hobby Chemist, not Professional or even Amateur
|
|
|
Mateo_swe
National Hazard
   
Posts: 541
Registered: 24-8-2019
Location: Within EU
Member Is Offline
|
|
Be aware that the thermometer in your drawing, if not secured enough can shoot down the condenser and break when using vacuum so make sure it cant
slip down.
Using an aspirator and a water pump can get medium vacuum and have many good properties.
There is no vacuumpump to damage with corrosive chemicals or solvents.
If using a waterpump to feed the aspirator you can just circulate the water so water consumption is none but any chemical coming over to the aspirator
goes into the circulating water (better than damaging the vacuumpump).
If putting ice in the waterbucket the vacuum is incresed, flow doesnt affect vacuum much as long its enough.
Its a cheap and good way.
Using a rotary vane vacuumpump can get you strong vacuum but chemicals get into the oil in the pump and the oil needs to be changed often.
Strong vacuum is quite dangerous, flasks can implode if not designed for high vacuum or if they have a small crack.
Chemical diaphragm vacuum pumps are designed to withstand chemicals and solvents, and are the best to use for chemical stuff but they are quite
expensive.
|
|
|
Sulaiman
International Hazard
    
Posts: 3692
Registered: 8-2-2015
Location: 3rd rock from the sun
Member Is Offline
|
|
So it circles back to the beginning,
the above description applies to the unit Yttrium2 first considered !
CAUTION : Hobby Chemist, not Professional or even Amateur
|
|
|
Cathoderay
Hazard to Self
 
Posts: 54
Registered: 29-1-2023
Location: US-Texas
Member Is Offline
|
|
This video gives more information.
https://www.youtube.com/watch?v=tYLlkTDstmo
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
Quote: Originally posted by Sulaiman  | Your diagram indicates that you want to do fractional distillations at low pressures.
I had such dreams too.
If there is a specific reduced pressure distillation that is very important to you, then go for it,
but I recommend that you work at atmospheric pressure only until you have a lot more practical experience,
by that time you will have a much better appreciation of what is required for your circumstances.
Not a criticism, just advice.
I'm still learning the hard way (mostly by failures) but I'm not in a hurry.
...........................
PS vacuum pumps for less than 10mmHg need not be expensive,
A peristaltic pump can be used for medium vacuum, low pumping rate applications
( my YT video https://m.youtube.com/watch?v=gGtYlS1ihbE)
A generic rotary pump (HVAC, car air-con service type) for medium vacuum at higher pumping rates
(either can give a gauge reading of - 1 Atmosphere)
I've not yet measured the actual minimum pressure of my rotary 
High vacuum is only for the few.
[Edited on 6-2-2023 by Sulaiman] |
Thanks for this! I was interested in seeing the peristaltic pump, -- I'm surprised the rollers do not tear up the tubing, but I guess they have less
friction than some other configuration.
I like your video!
You say high vacuum is only for the few, -- I'm still trying to understand why?
[Edited on 2/7/2023 by Yttrium2]
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
Quote: Originally posted by Mateo_swe  | Be aware that the thermometer in your drawing, if not secured enough can shoot down the condenser and break when using vacuum so make sure it cant
slip down.
Using an aspirator and a water pump can get medium vacuum and have many good properties.
There is no vacuumpump to damage with corrosive chemicals or solvents.
If using a waterpump to feed the aspirator you can just circulate the water so water consumption is none but any chemical coming over to the aspirator
goes into the circulating water (better than damaging the vacuumpump).
If putting ice in the waterbucket the vacuum is incresed, flow doesnt affect vacuum much as long its enough.
Its a cheap and good way.
Using a rotary vane vacuumpump can get you strong vacuum but chemicals get into the oil in the pump and the oil needs to be changed often.
Strong vacuum is quite dangerous, flasks can implode if not designed for high vacuum or if they have a small crack.
Chemical diaphragm vacuum pumps are designed to withstand chemicals and solvents, and are the best to use for chemical stuff but they are quite
expensive. |
Good point about the thermometer, how should it be affixed in vacuum distillation, -- are those ground glass thermometer thingy's the only option
here?
Do you have example of a flask not designed for high vacuum? (Aside from a star crack etc..)
Last point, -- You mentioned diaphragm vacuum pumps being expensive, in the video Cathoderay posted, it is stated that they're cheap? I'm sure there
is a range in prices for the different pumps but some clarification here?
[Edited on 2/7/2023 by Yttrium2]
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
Thank you everybody for chiming in 
I still have no idea of the range -- or is it level, in mmHg -- of vacuuum that the different pumps can produce.
Additionally I don't think <10mmHg is equivalent to 28 Inches Hg or higher, there must be a missing decimal point or something.

[Edited on 2/7/2023 by Yttrium2]
[Edited on 2/7/2023 by Yttrium2]
|
|
|
Cathoderay
Hazard to Self
 
Posts: 54
Registered: 29-1-2023
Location: US-Texas
Member Is Offline
|
|
I think there is some confusion about the way the amount of vacuum is stated.
There is absolute pressure that is measured from complete vacuum, so 10mmHg above absolute vacuum is a pretty strong vacuum.
760mmHg would be atmospheric pressure, no vacuum at all.
Another common way to express a vacuum, especially if it is a high vacuum is Torr which is pretty much the same as mmHg absolute. Very high vacuum is
very close to zero so it might be (10 to the minus 3) Torr. It is more and more difficult to get near zero. Mechanical piston or vane pumps can go
down to about 1mmHg. As shown in that video aspirator pumps cannot go down that low.
The other way to measure vacuum is gauge vacuum, it is measured from atmospheric pressure. Zero would be no vacuum. A complete vacuum would be
-760mmHg or about -30 inches Hg. Commonly if it is understood that you are talking about vacuum the "-" isn't used.
The bottom line is that the an aspirator is fine for filtering and distillation unless you want to distill oils or the like.
You don't have to be worrying about High Vacuum and Ultrahigh Vacuum and all that technology, you are not going to be distilling aluminum.
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
Quote: Originally posted by Cathoderay  | I think there is some confusion about the way the amount of vacuum is stated.
There is absolute pressure that is measured from complete vacuum, so 10mmHg above absolute vacuum is a pretty strong vacuum.
760mmHg would be atmospheric pressure, no vacuum at all.
Another common way to express a vacuum, especially if it is a high vacuum is Torr which is pretty much the same as mmHg absolute. Very high vacuum is
very close to zero so it might be (10 to the minus 3) Torr. It is more and more difficult to get near zero. Mechanical piston or vane pumps can go
down to about 1mmHg. As shown in that video aspirator pumps cannot go down that low.
The other way to measure vacuum is gauge vacuum, it is measured from atmospheric pressure. Zero would be no vacuum. A complete vacuum would be
-760mmHg or about -30 inches Hg. Commonly if it is understood that you are talking about vacuum the "-" isn't used.
The bottom line is that the an aspirator is fine for filtering and distillation unless you want to distill oils or the like.
You don't have to be worrying about High Vacuum and Ultrahigh Vacuum and all that technology, you are not going to be distilling aluminum.
|
I'm really lost now. I am able to take from this that there are two ways to measure vacuum.
Explain more please
And I take that I need rotary vane for oils and the like..
This is what I have so far,
Are these the two types of ways of measuring vacuum you were talking about ?
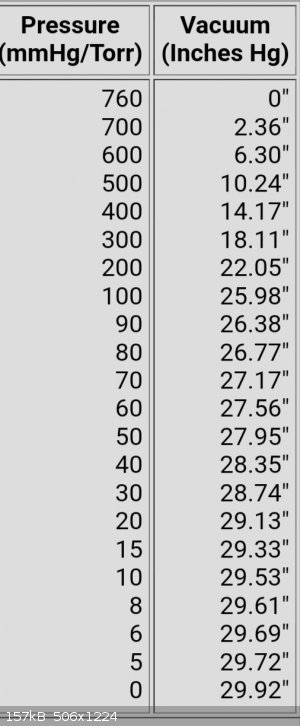
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
I heard this would be used for a vacuum fractional distillation as a coupling. I thought it had to be a ground glass thermometer w/th adapter
[Edited on 2/8/2023 by Yttrium2]

|
|
|
Cathoderay
Hazard to Self
 
Posts: 54
Registered: 29-1-2023
Location: US-Texas
Member Is Offline
|
|
There are many devices that can be used to measure a vacuum level.
I was talking about two basic ways of expressing the level.
There are many units to express the vacuum level.
This is a typical mechanical rotary vane vacuum pump.
https://www.harborfreight.com/25-cfm-vacuum-pump-61245.html?...
On the other hand this is a mechanical chemical resistant diaphragm vacuum pump.
https://www.southernlabware.com/oil-free-laboratory-chemical...
Do you notice a difference in price?
I don't know why you are asking so many questions about pumps.
Just use an aspirator!
Also there is not universal distillation set up, how it is built depends on what you want to do. How much vacuum level you would need depends on what
you want to do.
Maybe this explanation will help.
https://fluidpowerjournal.com/vacuum-measurement-a-basic-gui...
If you do some searching online you will find more info.
I have few questions for you.
What country are you in?
What level of schooling have you had?
I really don't have the time to cover an entire physics course about vacuum systems for you.
This is probably my last post on this thread.
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
Quote: Originally posted by Cathoderay  | There are many devices that can be used to measure a vacuum level.
I was talking about two basic ways of expressing the level.
There are many units to express the vacuum level.
This is a typical mechanical rotary vane vacuum pump.
https://www.harborfreight.com/25-cfm-vacuum-pump-61245.html?...
On the other hand this is a mechanical chemical resistant diaphragm vacuum pump.
https://www.southernlabware.com/oil-free-laboratory-chemical...
Do you notice a difference in price?
I don't know why you are asking so many questions about pumps.
Just use an aspirator!
Also there is not universal distillation set up, how it is built depends on what you want to do. How much vacuum level you would need depends on what
you want to do.
Maybe this explanation will help.
https://fluidpowerjournal.com/vacuum-measurement-a-basic-gui...
If you do some searching online you will find more info.
I have few questions for you.
What country are you in?
What level of schooling have you had?
I really don't have the time to cover an entire physics course about vacuum systems for you.
This is probably my last post on this thread.
|
More than happy to answer, I live in California. level of schooling, -- some community college with H.S. diploma... Got to general Chem 1, passed it.
A strong prealgebra background. (I hope) -- and some basic algebra skills. Its been years since my English writing classes, so I might be a little off
when it comes to the way things are worded, hopefully they are making sense.
I actually have almost everything complete at community college accept the chemistry, physics, math and foreign language. got a D in art, not sure if
that will transfer over ok, I can no longer audit it at one of my JC's...
And I've lurked and been here and around at other forums and such for the last however long since around 2005'
shroomery, wd, various automotive forums sciencemadness... --
Dad taught me a few things growing up'
Mainly, I probably wouldn't be into Chemistry if it wasn't for him.
Perhaps life has dealt me a different hand, perhaps I'm too lazy to learn -- Is that latter a logical proposition? One thing is for sure, I'm still
committed to learning chemistry and getting a degree. I spent the last 3 years on probation, moving from house to house, jail to jail, program to
program, out of my locale. It has been difficult having momentum during this times but I have managed to study, some, -- surely, not to the level that
I would have liked too.
Currently, I'd say I'm in Algebra1, General Chem1-- Although, I'll probably want to go back or something here, too.
I have about 3 straight years of math required for my degree, and foreign language. I'm bad at stacking classes with math. -- I will probably take 1
math class at a time and then physics / chemistry. -- Not sure when I'll study foreign language, it seems like it will really detract from learning
math chemistry and physics. If I take 1 course at a time, things might get stretched out too far and I might start forgetting by the time I'm done
with the foreign language, I might have forgotten some of the math? -- Or something? -- It has been this way with English, and perhaps a few other
subjects.
Anyways, that's a bit of my story and plan, to focus on solely math for as long as it takes. -- Even still, patience seems to be key for me...
|
|
|
Cathoderay
Hazard to Self
 
Posts: 54
Registered: 29-1-2023
Location: US-Texas
Member Is Offline
|
|
I wasn't after so much detail about your background.
If English wasn't your first language that might explain some difficulty following what I was saying.
The bit about probation and jail came as a surprise.
I have heard reports that the U.S. high school classes are poor in the sciences these days. That's sad because high school science should be the
foundation of further science education.
Perhaps you have been too focused on chemistry and missed some physics and other sciences. Of course mathematics is needed in any science pursuit.
You should try learning more on the internet on many sites and even books at the library. This forum cannot fill all your needs. I certainly don't
have time to answer all you questions, few people would.
I remember learning how barometers were made and worked when I was in high school. Maybe you weren't paying attention.
This site is good for some physics, interesting stories.
https://www.youtube.com/watch?v=oooS4Odq3PY
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
How many mmHg can this pull? It says .098Mpa
Or at what temperature can I boil water under full vacuum with this device?

Is this accurate?
How many mmHg does it pull!??? -- So at what temperature does water come over at this vacuum? -- I'm a little confused here, please elaborate, thank
you!!!!
[Edited on 2/21/2023 by Yttrium2]
[Edited on 2/21/2023 by Yttrium2]
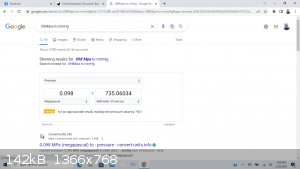
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
WE ARE AT 1 ATMOSPHERE HERE AT SEA LEVEL
1 ATM = 101325 PASCALS
101325 PASCALS
-98000 PASCALS (VACUUM LEVEL OF APPARATUS)
=
3325 PASCALS
= 24.93955 MILLIMETERS OF MERCURY.
= .98 inches of Hg
-Coincidence the vacuum level of the aspirator was .098 mpa?
Help I'm lost. EEEk!!!!
[Edited on 2/21/2023 by Yttrium2]
[Edited on 2/21/2023 by Yttrium2]
|
|
|
Yttrium2
Perpetual Question Machine
    
Posts: 1104
Registered: 7-2-2015
Member Is Offline
|
|
(rereading everything - one sec)
[Edited on 2/21/2023 by Yttrium2]
Ok so it still befuddles me how they figured out how much vacuum the vevor aspirator can pull?
[Edited on 2/21/2023 by Yttrium2]
|
|
|
| Pages:
1
2 |