The new One
Harmless

Posts: 5
Registered: 19-10-2018
Member Is Offline
|
|
Keto group hydrogenation of a-tertiary a-hydroxy ketones. Possible?
Hey chemical friends,
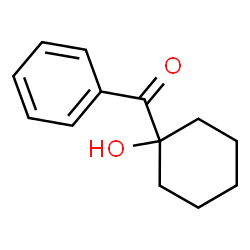
I've got some 1-Hydroxycyclohexyl Phenyl Ketone [Attached pic], it's fairly cheap and I hope to use it as PCA (Phenylcyclohexylamine) precursor.
The hydrogenated product (1-hydroxycyclohexyl)phenyl carbinol rearranged in hot dilute sulfuric acid to
2-Phenylcyclohexyl carboxaldehyde.
In cold concentrated sulfuric acid it rearranged mostly to Cyclohexyl Phenyl Ketone with the Carboxaldehyde as side product and no ring
expansion[Attached Pdf].
My first attemp to reduce the ketol with Sodium Borohydride in Isopropanol failed, no reduction takes place and the addition of water, acetic acid or
copper salts have no influence.
Next time I give Zinc borohydride a try.
Anybody else have an idea or know a proven procedure or about the impossibility?
Thx and have a nice Weekend.
TnO
Attachment: Ring-size-effects-in-the-pinacol-rearrangement.pdf (530kB)
This file has been downloaded 366 times
[Edited on 30-11-2019 by The new One]
|
|
|
zed
International Hazard
    
Posts: 2283
Registered: 6-9-2008
Location: Great State of Jefferson, City of Portland
Member Is Offline
Mood: Semi-repentant Sith Lord
|
|
Ummm. Not impossible, that a prolonged high temp heating in excess isopropyl alcohol might work. MPV?
Baring that, NaBH4/AlCl3, in diglyme might work.
OR........Maybe, just maybe, it is time to go to a good library, and search the literature. Somebody has done this before.
https://www.organic-chemistry.org/namedreactions/meerwein-po...
[Edited on 1-12-2019 by zed]
|
|
|
The new One
Harmless

Posts: 5
Registered: 19-10-2018
Member Is Offline
|
|
Have thanks for your response zed,
higher temperatures might work due to more activation energy, but about a-tertiary hydroxy ketones in the MPV, i've found only negative conclusions.
Before I check the activity of the Zinc Borohydride, i give it solventless at elevated temperatures a try, with and without different types of
catalyst.
Under Uv irradiation this compound forms benzoyl and cyclohexanol radicals.
Has Uv the ability to initiate the hydrogenation with borohydride, before formation of the reactive radicals take place?
Thx
TnO
[Edited on 1-12-2019 by The new One]
|
|
|
Cou
National Hazard
   
Posts: 958
Registered: 16-5-2013
Member Is Offline
Mood: Mad Scientist
|
|
One week in the library for every day in the lab.
|
|
|