JJay
International Hazard
    
Posts: 3440
Registered: 15-10-2015
Member Is Offline
|
|
Styrene Nomenclature
I looked around but didn't see a general thread on organic nomenclature, but if there is one, this probably belongs there.
I noticed recently that chemists seem to use different ordering rules for styrene than for other chemicals. The discussion on Ask Dr. Shulgin seems to
confirm this, but he is beating a strawman here: http://www.cognitiveliberty.org/shulgin/adsarchive/nomenclat...
I'm reluctant to dismiss Shulgin's statements as the rantings of a lunatic amphetamine chemist, but I'm not really seeing a lot of hard and fast rules
on prioritization of the alpha carbon in academic discussion of the topic: http://www.chem.ucla.edu/~harding/IGOC/A/alpha_carbon.html
Indeed, industry seems to use similar nomenclature: https://www.alfa.com/en/catalog/L03609/
What exactly are the rules on prioritization of the alpha and beta carbon?
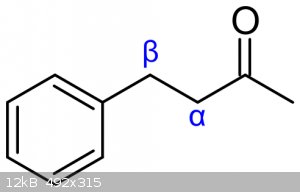 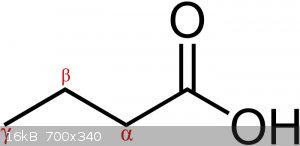
(Images found on Wikipedia.)
|
|
|
unionised
International Hazard
    
Posts: 5109
Registered: 1-11-2003
Location: UK
Member Is Offline
Mood: No Mood
|
|
Alpha is the next carbon along, beta is the one next to that.
The problem is where do you start.
In ring systems you (sometimes) start at the ring, and in ketones you start at the carbonyl.
It's usually apparent from context.
|
|
|
JJay
International Hazard
    
Posts: 3440
Registered: 15-10-2015
Member Is Offline
|
|
If you treat styrene as an alkene, things are a little more complicated than if you treat it as a ring system: https://socratic.org/questions/how-do-you-name-alkenes-and-a...
|
|
|
Melgar
Anti-Spam Agent
    
Posts: 2004
Registered: 23-2-2010
Location: Connecticut
Member Is Offline
Mood: Estrified
|
|
I guess you'd name styrene derivatives similar to, say, toluene derivatives. After all, the "-ene" at the end is used for aromatic rings, because
technically they're alkenes as well.
The first step in the process of learning something is admitting that you don't know it already.
I'm givin' the spam shields max power at full warp, but they just dinna have the power! We're gonna have to evacuate to new forum software!
|
|
|
JJay
International Hazard
    
Posts: 3440
Registered: 15-10-2015
Member Is Offline
|
|
Eh, alkenes don't have the same kind of resonance as aromatics, although there are some similarities. But come to think of it, styrene would have
resonance that isn't like just another alkene too....
|
|
|
Melgar
Anti-Spam Agent
    
Posts: 2004
Registered: 23-2-2010
Location: Connecticut
Member Is Offline
Mood: Estrified
|
|
Well, yeah. The point is that alpha, beta, etc. go away from the "main" or "defining" functional group. You can usually tell what the main
functional group is by looking at the suffix. However, structures like benzene, toluene, ethylbenzene, p-xylene, etc. have the benzene ring as their
main functional group. You'll notice that these all end in "-ene" too. Styrene is the same type of case, except that it also contains an aliphatic
alkene functional group. That doesn't really matter though, because its defining group is still the benzene ring. There's just confusion because
"-ene" is a suffix for both alkenes and benzene derivatives, I guess. But it still follows the pattern of all the other benzene-ring-containing
substituted hydrocarbons that end in "-ene".
The first step in the process of learning something is admitting that you don't know it already.
I'm givin' the spam shields max power at full warp, but they just dinna have the power! We're gonna have to evacuate to new forum software!
|
|
|
JJay
International Hazard
    
Posts: 3440
Registered: 15-10-2015
Member Is Offline
|
|
Yeah, I'm actually not sure how much it matters since it's a terminal alkene, but that resonance does explain why it is so easy to isomerize terminal
phenylpropenes to beta-methylstyrenes. Or I think so anyway. Science demands an answer 
|
|
|