quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
puzzled by Hg in H2SO4, oleum? not dissolved??? black precipitate, looks like elemental mercury
puzzled by a tiny bitty bead of mercury, under .116g (a larger bit i had side by side with it that i weighed earlier), maybe .59g or something that i
put in the acid layer from here
https://www.sciencemadness.org/whisper/viewthread.php?tid=41...
the acid was of unknown strength, although assumed high. in brief it was thought to be nitric and sulphuric both strong and have iodine in it,
possibly some iodic acid of gold, or rather HAuI4 (although this being a lewis acid? should have been extracted in the ether layer (lewis base)).
this was put in a small 10ml beaker with the tiny bead of mercury overnight and part of a days time to no apparent effect.
so this was put in a quartz tube and heated, with small UV lamps running on it. and the round ball broke up and appeared black like crystal growth
mixed with the non solidness of mercury (no picture taken at that time)
concentrated sulphuric acid was added ~10-15ml and it was run through the same process with the black part being a powdery appearence in one half
which did not conglomerate and another part that looked like a fused or burnt on bit (but it wasn't, it moved around, keeping its scabby like shape)
of mercury that was predominately burnt looking as was stated and blacked round the edges and near full through but in the center was seen to be a
silvery,non lustery (like when it makes amalgams) bit of mercury.
so this was heated higher and while i avoided it at first (wanting to keep nitrate/NO2 and I2 if it was there, to react with the Hg) ni heated it to a
higher temperature with UV and then after some time, removed the UV lamps, uncovered it and turned the heat even higher. and stopped heating as it had
nearly boiled down to maybe 5ml (from about 20ml). the fuming from the top was found to be acidic and the condensate around the top is a brownish
yellow (there is some whiteish/clear vapor condensate in the middle section). and the black mass is still on the bottom, a uniform clumped powder.
i am puzzled as i am guessing all the NO2 left, any I2, and that SO3 possibly and H2O as well were leaving. the vapor doesn't smell like SO2, and i
don't know the smell of SO3, although i thought it sort of smelt like NO2 with a bit of Iodine (i think that may be mental expectation).
so what i am guessing is that this is concentrated oleum and it still hasn't dissolved the mercury it seems. i don't know what double salt or salt it
could be if it isn't finely divided elemental mercury although i am mostly certain it isn't a single salt.
i am puzzled. what is this and why is it not dissolving in such concentrated acid?
edit: i did wash it with diethyl ether which seemingly should be in the acid to some degree. it has been sitting for about a month uncovered so i
don't know if there is any ether in it still. also the batch used was from the first wash and i originally thought it was the ether wash and attempted
to distill it over much to my frustration, only getting some liquid and the rest not boiling over, untill i realized it was the acid layer. if your
still with me, assuming there is no organics in this is a fairly safe guess, i guess and on with it. the pistures show the tube after i let it sit on
the hotplate as it cooled off, in turn boiling out about half of what was there from the 5ml and darkening it to the point that the pictures with this
camera are insufficient to see the black gunk on the bottom (i can barely see it through my goggles). the close up attempts to show there is some
purple blackish precipitate that recently formed as it boiled down, there is also some purplish fume it seems, over powering the brown in the tube
above the liquid. i am guessing this is iodine condensed and residual vapor, which i thought would have left the tube. it may be a salt/compound and
while i am curious to find about boiling near dryness, i don't want to ruin anything that may have formed. also i don't know about this oleum, if it
is oleum. the flask shown holds the remainder of the starting acid that i saved in it's original flask.
[Edited on 13-12-2014 by quantumcorespacealchemyst]
    
edit: also puzzlers, the acid layer i futily attempted to distill in the previus experiment, just after attempting to do that and transferring to
another flask. it has an odd nice looking golden hue which seemed (unsure) to have darkened/reddened over sitting (didn't take picture before
experiment, the picture of the remainder doesn't show it as well). the starting reaction was 5ml H2O, 2.602g KNO3, and 5.700gNaHSO4 boiled (to a
syrup) and condensed into a flask filled with iodine which was heated to fill it with vapor (idea was to see if iodine and SO2 making H2SO4 and HI
would work with NO3 to make a iodic aqua regia for the production of HAuI4 from gold). it didn't seem to work and the golden hued nitric acid mix and
iodine were eventually mixed with concentrated H2SO4 and run through a reaction that mostly dissolved all the gold (and redeposited about half that
dissolved in powder form {electrolysis was run on it while it heated and while i don't know if that is what helped the gold dissolved, it also
precipitated it out as a brown powder. [left the electrodes on after the heating too]}). so after washing with ether, that is the acid layer (should
have been rather concentrated, perhaps the iodine reduced the sulpur out of the acid and dissolved it in itself as it decomposed all the way down and
this isn't oleum and perhaps the mercury is mercury sulphide in it beta form (i believe)?)
please let me know what is most likely existing chemically here.
[Edited on 13-12-2014 by quantumcorespacealchemyst]
 
[Edited on 13-12-2014 by quantumcorespacealchemyst]
[Edited on 13-12-2014 by quantumcorespacealchemyst]
[Edited on 13-12-2014 by quantumcorespacealchemyst]
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
sulphur poly cations, mercury (and perhaps polycationic sulphur) pyrosulfates and hydrogen pyrosulfates
so it seems this may be it
"Sulfur polycations
Sulfur polycations, S8 2+, S4 2+ and S19 2+ are produced when sulfur is reacted with mild oxidising agents in a strongly acidic solution.[32] The
colored solutions produced by dissolving sulfur in oleum were first reported as early as 1804 by C.F Bucholz, but the cause of the color and the
structure of the polycations involved was only determined in the late 1960s. S8 2+ is deep blue, S4 2+ is yellow and S19 2+ is red.[10]"
http://en.wikipedia.org/wiki/Sulfur
[10] = Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. pp. 645–665. ISBN 0080379419.
[32] = Shriver, Atkins. Inorganic Chemistry, Fifth Edition. W. H. Freeman and Company, New York, 2010; pp 416
backups in case they are removed ie: acetyl chloride recently had it's full chemical equations of it's production removed to leave out the side
products, produced in the reaction, from being mentioned. hopefully this gets restored and the people editing the thing step down.
iodine being oxidizing i believe and any residual nitric acid seem to have done this with the H2SO4 and the SO3 absorbed by the H2SO4 producing oleum
and hopefully the S19+ polyanion, i haven't found any informations on anions of mercury/containing mercury and am currently guessing it is in it's
finely devided elemental form and/or in beta sulphide allotrope form buit this puzzles me.
"β-HgS is precipitated as a black powder when H2S is bubbled through solutions of Hg(II) salts.[6] β-HgS is unreactive to all but concentrated
acids.[4]
Mercury metal is produced from the cinnabar ore by roasting in air and condensing the vapour.[4]"
http://en.wikipedia.org/wiki/Mercury_sulfide
[4] Greenwood, Norman N.; Earnshaw, Alan (1984). Chemistry of the Elements. Oxford: Pergamon Press. p. 1406. ISBN 0-08-022057-6.
[6] Cotton, F. Albert; Wilkinson, Geoffrey; Murillo, Carlos A.; Bochmann, Manfred (1999), Advanced Inorganic Chemistry (6th ed.), New York:
Wiley-Interscience, ISBN 0-471-19957-5
so if there wasn't any H2S generated in situ (which i don't know), then there seems to be a different route it may have taken, but if it dissolves in
strong acids and it is in strong oleum, perhaps pure pyrosulphuric acid, then perhaps (assuming it isn't elemental mercury [which also puzzles me if
it is as it would seemingly react/dissolve in such strong acid]) there is so much sulphur dissolved that it is saturated, excluding the MgS from
dissolving. i believe Iodine can reduce as well as oxidize, although i am not sure, and possibly acted as both an oxidizer and reducer in the
reactions. also the smell above the mixture is of iodine.
edit: so i took a few more pictures as best i could and on rolling it around to get good pictures of the precipitate, there was a small swirl of
creamy whiteness, not unlike thick cream in coffee, i was unable to get a picture of it as it redissolved and it has me quite perplexed and somewhat
exited.
[Edited on 13-12-2014 by quantumcorespacealchemyst]
   
the discoloration on the work surface is from spilled chloroform extract of yerba mate, which made that odd texture and staining
[Edited on 13-12-2014 by quantumcorespacealchemyst]
"Sulfuric acid production
Oleum is an important intermediate in the manufacture of sulfuric acid due to its high enthalpy of hydration. When SO3 is added to water, rather than
simply dissolving, it tends to form a fine mist of sulfuric acid, which is difficult to manage. However, SO3 added to concentrated sulfuric acid
readily dissolves, forming oleum which can then be diluted with water to produce additional concentrated sulfuric acid.[1]"
http://en.wikipedia.org/wiki/Oleum
[1] Considine, Douglas M., Chemical and Process Technology Encyclopedia, McGraw-Hill, 1974, pp 1070–1.
and, under "As an intermediate for transportation"
"In addition, oleum is less corrosive to metals than sulfuric acid, because there is no free water to attack the surfaces.[2] Because of that,
sulfuric acid is sometimes concentrated to oleum for in-plant pipelines and then diluted back to acid for use in industrial reactions."
[2] http://www.sulphuric-acid.com/techmanual/Storage/storagetank...
which states verbatim " Metal loss increases as the acid strength decreases below 98% H2SO4. 77% H2SO4 is usually regarded as the practical lower
limit at which sulphuric acid can be stored at ambient temperatures in unprotected carbon steel tanks."
also though, reading further,
" Uniform Corrosion
The figure below illustrates the corrosion rate of steel by sulphuric acid as a function of concentration and temperature.
To compensate for uniform corrosion an adequate corrosion allowance has to be included in the design of the tank. It should be based on design life,
with consideration given to factors such as temperature, tank utilization and acid purity.
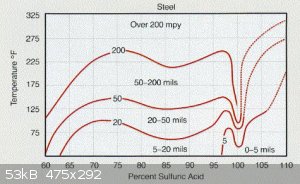
Temperature
The steel isocorrosion curves clearly illustrates that the corrosion rates increase with increasing acid temperature. Product acid from a plant
should be cooled to a maximum of 40°C (104°F). Within the storage tank the bulk acid temperature should be kept as low as possible without freezing
the acid.
Climatic conditions contribute to the temperature variation within a storage tank. Season temperature changes are generally not a problem because
rarely will the ambient temperature be higher than 40°C (104°F). Of greater importance is the effect of the sun heating the tank contents. Higher
corrosion rates have been reported on the side of tanks heated by the sun. To minimize this problems the tank should be painted with a heat
reflecting colour."
perhaps there is a pyrosulfate or hydrogen pyrosulfate of mercury produced here. what are the properties of (if they all
are known to exist) of Hg(S2O7), Hg2(S2O7), HHg(S2O7)? what about S19(S2O7)? besides the pyrosulfates, the solution looks more red than any of
the other dissolved polycations of sulphur in oleum, according to the top lines from wikipedia about them.
[Edited on 13-12-2014 by quantumcorespacealchemyst]
[Edited on 13-12-2014 by quantumcorespacealchemyst]
and wow, even though this is not what is made, as far as i know, this is amazing
"Logemann, Christian, Witt, Julia, Gunzelmann, Daniel, Senker, Jurgen and Wickleder, Mathias S. 2012, New Compounds Bearing [M(S2O7)3]2– Anions (M =
Si, Ge, Sn): Syntheses and Characterization of A2[Si(S2O7)3] (A = Na, K, Rb), A2[Ge(S2O7)3] (A = Li, Na, K, Rb, Cs), A2[Sn(S2O7)3] (A = Na, K), and
the Unique Germanate Hg2[Ge(S2O7)3]Cl2 with Cationic 1∞[HgCl2/2]+ Chains †, Journal of inorganic and general chemistry, vol. 638, no. 12, pp.
2053-2061. "
http://dro.deakin.edu.au/view/DU:30064494
https://www.researchgate.net/publication/264316009_ChemInfor...
and the Unique Germanate Hg2[Ge(S2O7)3]Cl2 with Cationic 1∞[HgCl2/2]+ Chains holy mol ee
"ABSTRACT The title compounds are prepared by reaction of MCl4 (M: Si, Ge, Sn) with oleum (65% SO3) and their crystal structures are determined by
single crystal XRD."
i wonder about H2O2 besides the danger, perhaps this leads to peroxydisulfuric acid/marshall's acid. i assume it is extremely exothermic and gas
producing but drop wise and cooled. i wonder about pyroselenic acid, H2Se2O7 and peroxydiselenic acid H2Se2O8.
[Edited on 13-12-2014 by quantumcorespacealchemyst]
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
.179g Ge and .393g powdered realgar ore, red, no yellow visable, seems quite pure, As4S4. added to mix, brought outside in cold night, set on clamp,
heated to red heat and fuming with propane torch. 3-5% H2O2 squirted in, washed down sides. heated again, frothing, tinkering, heating stuck on wall
bits as foaming rises, keeping foam from top. set to a decent boil, boiled mostly dry. foam and solution seemed cream,yellow orange light in
beginning, while solution became pale blue about half way to a third of the starting liquid level. the rest was observed from a distance to wait out
fuming and was reapproached when red heat seen in tube bottom. color not observed well, foggy goggles as well. reddish precipitate, slightly wetted by
condensate remnant. torch dryed bottom to top to drive off, clamp repositioned to get top part, mercury, it seems, condensed here (previous clamp
spot). white precipitate on bottom, dark grey part mostly seperated, white precipitate when tube brought indoors seemed to sublimate, unkown, seems to
have covered with a reddish hue so the bright white is a light/sparse red white, off white, to grey where blackish mass is. yellow and red precipitate
on tube walls, much like an orpiment covered realgar crystal (which decomposes. used to all be red like realgar and is mostly yellow save for the area
around where the clamp was, where the mercury, it looks, has condensed).
[Edited on 14-12-2014 by quantumcorespacealchemyst]
[Edited on 15-12-2014 by quantumcorespacealchemyst]
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
a bright white sublimate that sublimes on warming ~16°C from below freezing???
to save a read through, powdered Realgar, seems quite pure As4S4 [0.393g], Germanium powder [0.179g], added to
H2SO4 + HNO3, that had dissolved Iodine/Iodine compound (slim possiblity of HAuI4) that had a bead of mercury in it [~0.59g] , [~5-10ml] with more
H2SO4 added [~15-20ml] to help it dissolve. all boiled down to a brown liquid [~2ml] (brown fumes and white/clear fumes, condensates in tube wall,
rise from tube)
In quartz tube, heated with propane torch to red heat in temperature below freezing. fumes. 3-5% H2O2 [~60-70ml] added to wash down tube walls. boiled
off (dilute yellow/orange turns to pale blue at at least 1/2 liquid volume).
red coating, wetted, on tube walls, white precipitate on bottom [grey bit towards center where a chunk was not red heating as much as it was jutting
up from the surface during the reaction].
heated with strokes from bottom to top to drive off moisture. red turns yellow mostly. moved clamp location to get that spot with torch, and when
finished, that area has shiny dark black beads and red borders before the yellow decribed.
brought out of cold, white shiny powder seems to dissapear on rechecking it over a few to ten minutes while warming towards room temperature of around
16°C.
quite puzzling
i am convinced now that the white powder i saw on observing after fusing was indeed a sublimate [at cold toward room temperature]. the powder from the
bottom of the tube did look less than the copious white mass i thought i saw when observing it as cooling. i thought there was a bit less bright white
powder after i looked at the tube and then checked back after moving something/shuffling stuff, as it was warming up from the cold outside. i thought
about it quick, put it back, left it sitting to go do another quick task and on re-observing, there was no white powder in the tube. i figured, the
stuff on the bottom that was off whitish, red looking with grey bits was it after doing a surface reaction (the tube wall on the outside was still
white as seen in pictures), but it puzzled me that it seemed to be less mass significantly, actually volume, less volume as i don't know the mass.
about 1/2 less volume.
i didn't take a picture of the powder collected to a stoppered flask as the camera is not very good at that kind of detail, no matter how i attempt to
get fine detail in these logs. if i can get one, then i may post it. the powder on observing is about half medium to light grey and very light
purplish pink [the kind that seems a mix in with a white substance]. on scraping at the last bits from the tube [greyish part coating fused partly to
the tube] i noticed it was slightly wetted when brushing it off in to the flask [the first peices seemed dryer]. i dont know if this is moisture (may
be arsenic pentoxide, As2O5 and if hydroscopic if like P2O5) or mercury amalgam (uranium oxides reportedly amalgam and they arent pure metal so i am
guessing this can happen with many other substances). more realistically, i believe it is water absorbsion, as when i washed the outside of the tube
with warm to hot water, it seemed to have water vapor condense on the inside tube walls. i am guessing it is partly As2O5.
http://en.wikipedia.org/wiki/Arsenic_pentoxide
"Arsenic pentoxide can be crystallized by heating As2O3 under oxygen. This reaction is reversible:[2]
As2O5 <---> As2O3 + O2
Strong oxidizing agents such as ozone, hydrogen peroxide, and nitric acid convert arsenic trioxide to the pentoxide.
Arsenic acid can be generated via routine processing of arsenic compounds including the oxidation of arsenic and arsenic-containing minerals in air.
Illustrative is the roasting of orpiment, a typical arsenic sulfide ore:[6] 2 As2S3 + 11 O2 → 2 As2O5 + 6 SO2"
a bright white sublimate that sublimes on warming to the room temperature of ~16°C from below freezing. is
puzzling to me, and quite amazing, what do you think it is?
[Edited on 15-12-2014 by quantumcorespacealchemyst]
[Edited on 15-12-2014 by quantumcorespacealchemyst]
|
|
|
forgottenpassword
Hazard to Others
  
Posts: 374
Registered: 12-12-2013
Member Is Offline
Mood: No Mood
|
|
From the outset you have been using impure and contaminated substances, mixing them haphazardly, and seemingly doing processes without any knowledge
or expectation of the result. What is the purpose? If you are unable to follow the course of your reactions, and have no clue as to what you have
made, where is the sense in it?
Why do you start out by mixing mercury with nitric and sulphuric acids with iodine and (possibly) some HAuI4 dissolved in it. From the very start you
have NO IDEA of what is in your reaction mixture. Maybe there is some HAuI4, maybe there isn't. Maybe it would have reacted in your further
processing. But maybe it wasn't even there to start with. It is a waste of time.
And now you expect anyone to tell you what you have made?! You don't even know what reactants you've used! Since it is painfully obvious that you have
no education in chemistry whatsoever, may I suggest that you work with less toxic compounds than you are currently doing. If by some fluke you have
made a volatile mercury compound (or if you shall do so in the future), then your life-expectancy will not be very long at all. I don't expect for one
minute that you shall heed this warning, but it makes me feel better to have given it. I'll let nature take it's course: but nature punishes stupidity
and ignorance rather harshly; so I'll hope for the best for you, instead, and hope that you have unnaturally good luck.
[Edited on 15-12-2014 by forgottenpassword]
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
some person who can think of a highly volatile/sublimating white powder that may arise from the above described in my last post. please let me know
what it may be.
i am guessing the mercury is elemental or amalgamated with germanium a bit.
all the sulphur didn't leave but the powder obtained may not contain much of it. there seems to be iodine being the purplish hue and the reddish hue i
am guessing has sulphur as As4S4. it seems it is mostly arsenic oxides possibly mostly pentoxide with trioxide possibly contamininate but i doubt to
any degree as there was much heating and oxidants. the grey and the blackish mercury is what i wonder about as well as the white pwder that
dissapeared. i am guessing there is germanium involved in the grey and black stuff, not sure, hopefully something interesting. the idea from the paper
in the second post of mine was half used and decided to throw in a curve ball as the starting point of the 2ml brown liquid was an unknown and got no
ideas about it so instead of discarding, on it went. i don't know what that brown liquid was but it sure is interesting being a small amount of
elements being involved and some weird mechanics unknown to me taking place.
the white may be GeS2 that was fluffy and decomposed to GeS reducing volume and becoming greyish, reddish. so no super mystery i guess,
[Edited on 15-12-2014 by quantumcorespacealchemyst]
|
|
|
MrHomeScientist
International Hazard
    
Posts: 1806
Registered: 24-10-2010
Location: Flerovium
Member Is Offline
Mood: No Mood
|
|
You're all over the map here. Gold, mercury, iodine, germanium, arsenic, sulfur, all thrown together haphazardly with no discernible rhyme or reason.
All that plus impure reagents where you have no idea what is in them. As forgottenpassword asked, what is the purpose of all this? If you have no idea
what you are doing mixing things at random, then you're bound to have no idea what products you'll end up with. Working with scary stuff like mercury
and arsenic compounds, this is the last thing you want to be doing. Who knows what kind of volatile toxic compounds you might create?
Slow down, take some deep breaths, and start over. Come up with a plan. Write down what you think will happen. Document your steps. You must follow
the scientific method if you want to do science. If you can't make your experiments repeatable, there isn't much point.
Garbage in, garbage out.
[Edited on 12-17-2014 by MrHomeScientist]
|
|
|
phlogiston
International Hazard
    
Posts: 1378
Registered: 26-4-2008
Location: Neon Thorium Erbium Lanthanum Neodymium Sulphur
Member Is Offline
Mood: pyrophoric
|
|
Quote: Originally posted by quantumcorespacealchemyst  | this was put in a small 10ml beaker with the tiny bead of <b>mercury</b> overnight and part of a days time to no apparent effect.
so this was put in a quartz tube and <b>heated</b>, with small
-snip-
SO3 possibly and H2O as well were leaving. <b>the vapor doesn't smell</b> like SO2, and i don't know the smell of SO3, although i
|
So, you added mercury, heated it and then smelled it??
I hope this is not what causes you to post pages and pages of chaotic experiments with no clearly identifiable purpose.
I couldn't be bothered to read any further but casually noticed you experiment with arsenic compounds as well. You may want to reconsider if that is a
smart idea.
<b>On second thought, does anyone else feel this might be another reincarnation of our village idiot, with another "xyzChemist" name, that has
adopted a new style?</b>
[Edited on 17-12-2014 by phlogiston]
[Edited on 17-12-2014 by phlogiston]
-----
"If a rocket goes up, who cares where it comes down, that's not my concern said Wernher von Braun" - Tom Lehrer |
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
main thread title is main question, re testing without possible gold salts and/or iodine inclusions in starting acid not done yet, please note bottom
highlighted text
Quote: Originally posted by MrHomeScientist  | You're all over the map here. Gold, mercury, iodine, germanium, arsenic, sulfur, all thrown together haphazardly with no discernible rhyme or reason.
All that plus impure reagents where you have no idea what is in them. As forgottenpassword asked, what is the purpose of all this? If you have no idea
what you are doing mixing things at random, then you're bound to have no idea what products you'll end up with. Working with scary stuff like mercury
and arsenic compounds, this is the last thing you want to be doing. Who knows what kind of volatile toxic compounds you might create?
Slow down, take some deep breaths, and start over. Come up with a plan. Write down what you think will happen. Document your steps. You must follow
the scientific method if you want to do science. If you can't make your experiments repeatable, there isn't much point.
Garbage in, garbage out.
[Edited on 12-17-2014 by MrHomeScientist] |
yes and no, i comprehend and agree with you and also have the idea of tinkering just to see. the first case starts from a known, to a degree acid,
sulphuric, with most certainly, nitric in it. the added unknowns are possibly iodic acid of gold, that seems to be all. one may think of it as a spice
that may change the plate completely, or do absolutely nothing.
the reasonable thought here is there is no methyl/organic mercury and while inorganic compounds of mercury may be harmful, they are most all [as far
as i have found] cumulative toxins, needing quite a bit to be acute and are rid by the body over time. considering that this is minute quantity of
metal being worked with. the risk is to me is within a safe range.
i am wondering here, 1 why did mercury not dissolve so well, if at all? 2 why did the acid volume boil down without seemingly emitting vapor and become brown and slightly fuming, even to near
nothing?these question are asked in above posts with speculations leading to, 3
are there polyatomic cations of sulphur responsible for this reduction without so much fuming?
thanks
while i want to have pure reagents, i also want to know how to purify the impure ones
[Edited on 18-12-2014 by quantumcorespacealchemyst]
[Edited on 18-12-2014 by quantumcorespacealchemyst]
|
|
|
MrHomeScientist
International Hazard
    
Posts: 1806
Registered: 24-10-2010
Location: Flerovium
Member Is Offline
Mood: No Mood
|
|
With the number of possible impurities so high, and the large number of different things you did during the experiment, it's very difficult to answer
any questions here. Reduce the number of variables, start with known quantities and purities, try one thing at a time, and you can start to
investigate what happened. Put mercury in lab grade sulfuric and nitric acids. Does it dissolve? If yes, try again and this time add "iodic acid of
gold" or whatever you think was in your contaminated acid mix. Like I said before, try to reproduce your results and approach it in a more systematic
manner. In my opinion, 'tinkering' with chemistry is a very dangerous prospect, especially with what you're dealing with.
Maybe others would like to take a stab at it, but frankly I'm overwhelmed.
Also I hope you're disposing of these mystery toxic-salt-containing liquids properly!
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
solid form, i save most everything as i usually wish i had it later. while i want to know how to purify and reuse, there is a hazardous waste
collection i need to look into and/or where the local school labs dispose of their reagents.
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
I realize when I wrote solid form, it doesn't make apparent sense. What I mean is the stuff is all in solid form. Since then I have taken the
purpleish dark greyish material, in a mini erlimeyer and added some 5-10ml distilled H2O. It has settled mostly and I am curious to know what is
dissolved.
|
|
|
woelen
Super Administrator
        
Posts: 8003
Registered: 20-8-2005
Location: Netherlands
Member Is Offline
Mood: interested
|
|
I would suggest you to discard all of it and bring it as heavy metal waste to a proper municipal waste processing facility.
If you want to do new experiments, then try to be clear in what you do and also make clear what the purpose of the experiments is. The purpose may be
something academic (e.g. investigating complexes of a certain metal), or something practical (e.g. making and isolating an interesting compound), but
you MUST have a purpose. Otherwise you are just wasting time and materials.
|
|
|
Loptr
International Hazard
    
Posts: 1348
Registered: 20-5-2014
Location: USA
Member Is Offline
Mood: Grateful
|
|
Yeah, this is just asking for trouble. If you can get the solid into solution, you might be able to precipitate the heavy metals as their hydroxide or
carbonate. Then at least you may have recovered *some* of your starting materials, because who knows what could have taken place in the vessel.
EDIT: Wait, you say you use oleum, but then question if it is oleum!!?
EDIT: I think you are staying true to your username: alchemy. I looked at a couple of other posts, and I see a occurrence of the heavy metals listed
here. What are you trying to accomplish?
[Edited on 12-2-2015 by Loptr]
|
|
|
Texium
Administrator
       
Posts: 4554
Registered: 11-1-2014
Location: Salt Lake City
Member Is Offline
Mood: PhD candidate!
|
|
You know, just looking at all of these threads started by QCSA make me really sad. So many interesting and expensive reagents, and oftentimes nice
glassware too, all just thrown together in such an irresponsible way. I wish that I could afford to experiment with gold, mercury, geranium, and
iodine... I certainly wouldn't do crap like this.
QCSA, you should honestly take all of your expensive reagents and lock them up until you can figure out how to use them responsibly, and then,
actually do some real chemistry that will actually receive some praise and recognition instead of confusion and contempt. I can understand the feeling
you have of wanting to discover something new and thinking you can go about it with random conjecture and experimentation, but that doesn't work.
You've got to show some restraint and figure out the basics first. Before doing any more chemistry with gold and other expensive metals, I'd highly
recommend that you start with cheaper and more well-characterized elements like copper and iron.
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
I don't need praise and recognition. I need help doing cool stuff.
[Edited on 18-2-2015 by quantumcorespacealchemyst]
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
I don't like that Texas restricts chemists from owning specialized glassware. It is unjust.
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
This is the evaporated crystals from the water wash of the substance from the bottom of the tube. The flask on the right now has ~4 times more water
in it. There is an extremely tiny amount of compound that was soluble, as seen in the photo. I am guessing Arsenic acid possibly. It is mostly white,
a little grey on the edges made its way in.
So this impure compound in the flask seems to be fairly insoluble in water.

|
|
|
Texium
Administrator
       
Posts: 4554
Registered: 11-1-2014
Location: Salt Lake City
Member Is Offline
Mood: PhD candidate!
|
|
What you're not seeing though is that you
aren't doing cool stuff. You're making a very expensive mess.
True, but that
has nothing to do with it...
|
|
|
j_sum1
Administrator
       
Posts: 6277
Registered: 4-10-2014
Location: Unmoved
Member Is Offline
Mood: Organised
|
|
I concur. However, note that this is a metaphor. I suspect there are real issues with breathing deeply in your lab.
|
|
|
quantumcorespacealchemyst
Banned Shitposter
Posts: 213
Registered: 17-10-2014
Member Is Offline
Mood: No Mood
|
|
it tuns out, the H2SO4 i was using was 2N, nowhere near the 98% o 95% i thought i had (wrote one o those somewhere). that explains my constant
puzzlement about it's peculiar properties and failed reactions.
|
|
|
|