
MattEx - 3-8-2007 at 17:01
Anyone know a procedure for adding a carboxylic acid group to the right position of 1,3,5-cycloheptatriene to yield the named target product  ?
?
chemrox - 3-8-2007 at 19:15
Is that named correctly? I would have thought 2,4,6-cycloheptatriene carboxylic acid or 2,4,6-cycloheptatriene-1-carboxylic acid would be better
names. However, I am no IUPAC expert by any means. Now you have an addition or substitution reaction to a conjugated cyclic alkene. Can you Google
some information about the next step?
Here's a good starting place:
http://www.cem.msu.edu/~reusch/VirtualText/intro1.htm
JohnWW - 3-8-2007 at 20:11
2,4,6-cycloheptatriene fairly readily loses an hydride ion, e.g. by reaction with a sufficiently stable carbonium cation like the triphenylmethyl
cation, to become the resonance-stabilized planar tropylium cation, C7H7(+). So any reaction procedure involving an intermediate in the form of a
carbonium cation, in this hypothetical case (+)COOH, is liable to have the same effect
Leopata - 6-10-2011 at 22:10
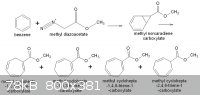
Benzene + methyl diazoacetate = methyl cycloheptatriene carboxylic acid methyl ester, via norcaradiene methyl ester.
The corresponding norcaradiene carboxylic acid, at high temperatures rearranges with ring expansion to the cycloheptatriene carboxylic acid methyl
ester, an important reagent to 2-tropene-2-carboxylate ring. I don´t have the literature at hand now, but with this you can start your investigation.
Hope you have what you were looking for.
Leopata - 6-10-2011 at 22:15
Here´s the reaction scheme
Leopata - 6-10-2011 at 22:29
Sorry...there´s a mistake in the figure above.
[Edited on 8-10-2011 by Leopata]
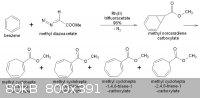
Retard-3000 - 8-10-2011 at 13:43
What do you want this 2,4,6-cycloheptatriene-7-carboxylic acid for ?

 ?
?