
TheMrbunGee - 22-7-2018 at 04:51
Hey SM,
Today I come to You with another mystery solid.
This should not be hard!
Do I got this from the same institute of Inorganic chemistry.
It is shaped like a parallelogram from the front and back and rectangle from all other sides. I got it in two forms, 2 mm and 4 mm high, and both 25
mm deep and 50 mm wide.

It is red-orange transparent solid with pretty low melting point. ( I thought it was just some kind of glass and for safety tried its compatibility
with heat) it melts @ about 100-200 °C (I can test for more accurate MP, if needed. )

when in gas flame, it gives bright blue color flame and some of solid boils and condenses as a yellow layer on solid itself.
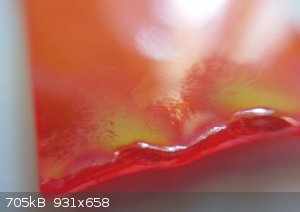
It has density of about 7.35 g/ml, it feels quite heavy.
It has these markings on the side.

My guess was tin iodide, but it is much more dense.
Do you have any guesses? Any tests in mind?
BTW: it is not soluble in water. and it is not hard at all.
[Edited on 22-7-2018 by TheMrbunGee]
[Edited on 22-7-2018 by TheMrbunGee]
unionised - 22-7-2018 at 05:40
STOP SETTING FIRE TO RANDOM THINGS.
It's almost certainly KRS 5 which is remarkable toxic.
http://www.internationalcrystal.net/optics_17.htm
https://www.spectral-systems.com/products/krs-5-atr-optical-...
TheMrbunGee - 22-7-2018 at 06:32
Yeah, I probably should! Good thing I had strong ventilation on, when I was heating it.
Looks like it really is Thallium Bromo-Iodide.
Well - I have been touching it bare handed, so I should have some prussian blue for dinner.
And I told it would be easy.  not sure how did I underrated the melting point
so badly.
not sure how did I underrated the melting point
so badly.

 not sure how did I underrated the melting point
so badly.
not sure how did I underrated the melting point
so badly.