
I really like these puzzles they should be done more often.
[Edited on 11-4-2013 by Boron Trioxide]



 Or pure coniine?
Or pure coniine?


 thionyl chloride? Triethylborane? Diphosphane?
thionyl chloride? Triethylborane? Diphosphane? 



Quote: Originally posted by Boron Trioxide  |


Quote: Originally posted by Endimion17  |






 You're the only one who has
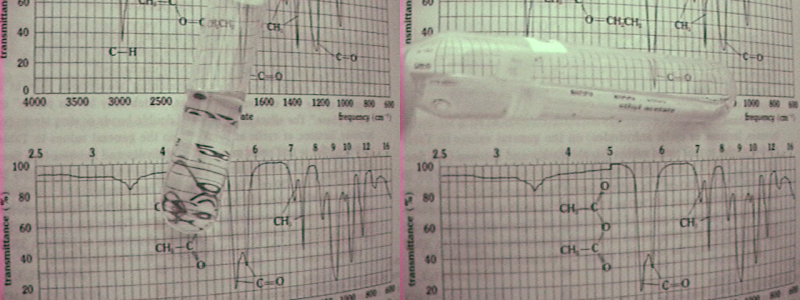
recognized the infrared spectrum table behind. It was one of the clues. Nice work.
You're the only one who has
recognized the infrared spectrum table behind. It was one of the clues. Nice work. 
Quote: Originally posted by woelen  |
Quote: Originally posted by Finnnicus  |

Quote: Originally posted by Pok  |

Quote: Originally posted by Finnnicus  |
Quote: Originally posted by blogfast25  |
Quote: Originally posted by Finnnicus  |
Quote: Originally posted by Pok  |
Quote: Originally posted by neptunium  |

Quote: Originally posted by Finnnicus  |
Quote: Originally posted by Finnnicus  |
Quote: Originally posted by Finnnicus  |
Quote: Originally posted by Endimion17  |
Quote: Originally posted by DraconicAcid  |









Quote: Originally posted by Pok  |
| Quote: |


Quote: Originally posted by woelen  |
Quote: Originally posted by blogfast25  |

Quote: Originally posted by Eddygp  |
Quote: Originally posted by Endimion17  |



Quote: Originally posted by Eddygp  |
Quote: Originally posted by garage chemist  |