
Is it possible to react 1,1-2,2 tetrachloroetylene with AgNO3 ? or other similar product ? what solvent is best suited for this ?

Quote: Originally posted by virgilius1979  |
Quote: Originally posted by Motherload  |
Quote: Originally posted by virgilius1979  |
Quote: Originally posted by Motherload  |
Quote: Originally posted by APO  |
Quote: Originally posted by virgilius1979  |
Quote: Originally posted by APO  |
Quote: Originally posted by AndersHoveland  |

Quote: Originally posted by AndersHoveland  |
Quote: Originally posted by DraconicAcid  |
Quote: Originally posted by AndersHoveland  |
Quote: Originally posted by KonkreteRocketry  |






Quote: Originally posted by Motherload  |
Quote: Originally posted by Motherload  |
Quote: Originally posted by KonkreteRocketry  |
Quote: Originally posted by KonkreteRocketry  |
Quote: Originally posted by woelen  |
Quote: Originally posted by APO  |
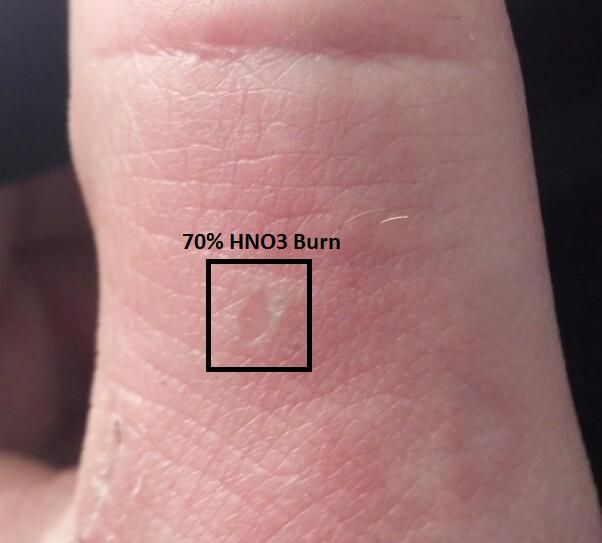
Quote: Originally posted by APO  |
Quote: Originally posted by APO  |
Quote: Originally posted by APO  |
Quote: Originally posted by APO  |
Quote: Originally posted by APO  |
