
pyrofrench - 2-10-2002 at 10:05
hello everyone i'm a french pyro.
i'am 19, i leave nearby to paris.
my question: is it possible to make camphor peroxide ?
acetone is a keton
MEK is a keton
and camphor as well so why not.
sorry, i speak not very well english !
thank you for your answer.
PS in french there is only one forum "pyro"
[Edited on 28-7-2004 by chemoleo]
NERV - 2-10-2002 at 10:18
I think that it is possible to make camphor peroxide. But I may be wrong as my chemistry isnt all that good.
Nick F - 2-10-2002 at 11:14
I've thought of it in the past, but when you look at the structure of camphor you'll see that the OB would suck, and I think the peroxide would be
even less stable than most, if preparable.
BLAST_X - 3-10-2002 at 13:30
You should test to oxidize synthetic camphor to form a peroxide or a hydroperoxide.
bornane-2-one
1,7,7-trimethylbicyclo(2.2.1)heptan-2-one
C10H16O
I doubt its
Boob Raider - 20-10-2002 at 17:17
going to work. Camphor ain't soluble in H2O. I don't think peroxidation works well in alcohol, and if you use acetone, you'll end up with AP. But as
of now an alcohol soln is worth a try. Although if you boil camphor in conc HNO3, you will probably get nitro camphor or something which you can again
try to peroxidize.
Camphor
trillian - 23-10-2002 at 02:28
Camphor are a very varied usable compound.
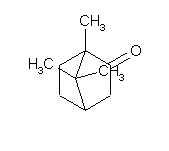
Does exist camphor peroxide?
kazaa81 - 1-5-2004 at 12:38
Hallo,
I'm doing a research about the ketone camphor and I need to know if it can formate peroxides. If anyone can help me, saying yes or no and, if
yes, the equation.......this time Google haven't helped me.
Thanx 
terpene peroxides
Polverone - 27-7-2004 at 19:59
If it is unsaturated (with a C=C double bond), a terpene should certainly be capable of forming a peroxide. Camphor (which contains a keto group) does
not meet this requirement, but camphene, which contains a C=CH2 group, does.
An example is cineole, a major constituent of eucalyptus oil, which is a naturally occurring terpene peroxide, and indeed probably largely responsible
for its antiseptic properties. I believe it, or a similar compound, occurs in teatree oil (either melaleuca (Australia) or leptospermum (New Zealand)
oil), which is also antiseptic. I remember reading somewhere that an industrial process for manufacturing hydrogen peroxide was, and may still be,
based upon the synthesis and reaction of a terpene peroxide.
John W.
(this was actually written by JohnWW but accidentally "reported" instead of "replied"
unionised - 28-7-2004 at 13:25
The discussion on camphor peroxide indicated that it would suck as an explosive and that it would be difficult to make, but that doesn't rule it
out.
Acetone is a bit short on carbon-carbon double bonds, but I hear it forms a peroxide.
IIRC tea tree oil is largely terpineol; it might be possible to replace the OH with OOH but it wouldn't really be on- topic.
Last time I checked, the industrial production of hydrogen peroxide used the oxidation and reduction of a substituted anthraquinone.



