vano - 31-12-2020 at 08:02
Today I made Gallium (II) iodide. It is a green solid. I tried this particular method:
Ga + HgI2 = Hg + GaI2
I did not try to separate the compound, I just wondered if it was real. However, to some extent the compound will be contaminated with mercury. I
think it is an interesting and simple reaction.

Metallophile - 31-12-2020 at 10:10
Wikipedia says this is a mixed valence compound of Ga(I) and Ga(III). This page on "GaI" is pretty interesting:
https://en.wikipedia.org/wiki/Solid_%22GaI%22_Precursor
vano - 31-12-2020 at 10:40
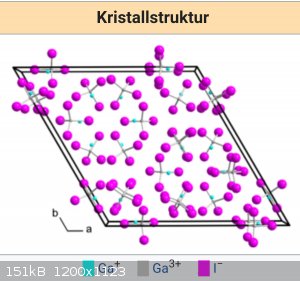
Yes it is a mixed compound. Gallium 1 and 2 iodides two of them are green. Look at crystal structure. But i like this reaction, because its simple and
this compound is very nice and interesting.