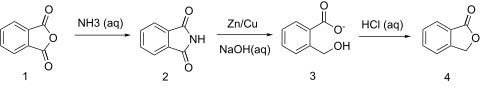
That's a very interesting synthesis you've posted. Apparently, yes, you can do the same thing with succinic acid. The reaction with ammonia, at least,
to form succinimide.
https://thosci.com/synthesis-of-suiccinimide/
Unrelated to the topic at hand, but from there, you can brominate the succinimide in an aqueous solution with NaOH to yield NBS. Potentially a very
useful pathway, from OTC chemicals.
I was interested more in the reduction of succinic acid to yield THF, but if the pathway you've posted holds up for succinic acid, it may be an easily
accessible option for GBL.
edit:
I can't imagine it would be all that hard to construct a suitable pressure vessel for containing such a reaction. The biggest issue would be
pressurizing a hydrogen atmosphere. I don't know how you could do this outside of an external tank. Perhaps you could generate hydrogen gas in some
sort of sealed container (like the NaOH + Al method). I'm imagining a pressure cooker type system with an attached pressure gauge. You could attach
some sort of ball valve and gas line to the reaction vessel, and pressurize it.
You'd still need some way to flush the system with nitrogen, or risk turning your steel reaction vessel into an IED. Nitrogen tanks are fairly cheap I
imagine. It seems fundamentally simple though. So long as you take appropriate precautions, you could easily hydrogenate effectively anything,
forever, in a batch-wise process. So long as you have a catalyst, of course.
Am I wrong in this assumption?
[Edited on 9-5-2019 by advanced warning] |