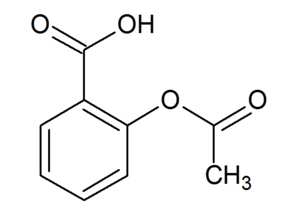
Acetylsalicylic acid
Acetylsalicylic acid is an organic acid commonly known as aspirin.
Acetylsalicylic acid (ASA) is prone to hydrolysis, breaking down into salicylic acid and acetic acid. Solutions of ASA are therefore not very stable and hot or boiling water will quickly break apart the molecule into the two smaller organic acids. The solid must be protected from air as moisture in the air will also degrade it.
Hydrolysis of ASA can be observed through the smell of acetic acid, solutions that have undergone significant hydrolysis have a very noticble vinegar smell to them.
Availability and preparation
As one of the world's most commonly used medicine, aspirin is found in every pharmacy on earth as well as most supermarkets. Even basic aspirin tablets contain some form of binder to keep it in tablet form or make it more air stable. To remove these, the tablets can be crushed and washed with denatured alcohol. The ASA is soluble in the alcohol while the binders are not, so any solids that remain undissolved can be filtered off.
The alcohol can then be carefully boiled off or evaporated away. Acetylsalicylic acid forms light, fluffy, glittering white crystals when recrystallized from solution. These crystals should be stored in a dry, airtight container to prevent hydrolysis. Some hydrolysis to salicylic acid and acetic acid is unavoidable, however, so acetylsalicylic acid cannot be kept for very long periods of time without loss in purity.
Reactions
Acetylsalicylic acid readily hydrolyzes in hot water or solutions that are highly acidic or basic. While this can be an obstacle for some syntheses, it can be taken advantage of to make the more stable salicylic acid, which fills many of the same roles, by refluxing acetylsalicylic acid powder in low pH solution, cooling, and filtering out the crystals of salicylic acid, which should be recrystallized.
ASA reacts with sodium carbonate and bicarbonate to form sodium acetylsalicylate. Sodium hydroxide cannot be used to make the sodium salt as it will hydrolyse the ASA.
Purified aspirin is the precursor in one of the most commonly used syntheses of trinitrophenol (picric acid), a dye and explosive.
'Copper aspirinate' can be formed by using the ASA as a ligand to the copper(II) ions, or simply precipitated by the mixture of solutions of copper(II) sulfate and sodium acetylsalicylate. This is discussed in detail at http://www.sciencemadness.org/talk/viewthread.php?tid=9920