HMX
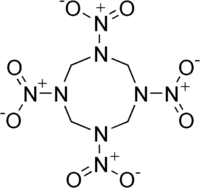 HMX structure
| |
| Names | |
|---|---|
| IUPAC name
1,3,5,7-Tetranitro-1,3,5,7-tetrazoctane
| |
| Other names
Cyclotetramethylene tetranitramine
Cyclotetramethylenetetranitramine Her Majesty's Explosive High Melting Explosive High-Molecular-weight RDX High-velocity Military Explosive Octogen Octahydro-1,3,5,7-tetranitro-1,3,5,7-tetrazocine | |
| Properties | |
| C4H8N8O8 | |
| Molar mass | 296.155 g/mol |
| Appearance | White colorless solid |
| Odor | Odorless |
| Density | 1.91 g/cm3 (25 °C)[1] |
| Melting point | 276–286 °C (529–547 °F; 549–559 K) |
| Boiling point | 287 °C (549 °F; 560 K) Decomposes |
| 0.000334 g/100 ml (20 °C) 0.000446 g/100 ml (25 °C)[2] | |
| Solubility | Solvates in DMF, NMP Soluble in butyrolactone, dimethyl sulfoxide Slightly soluble in acetone, acetonitrile, cyclohexanone |
| Solubility in acetone | 2.8 g/100 ml (25 °C) 4.2 g/100 ml (56 °C) |
| Solubility in acetonitrile | 2 g/100 ml (25 °C) 7.3 g/100 ml (82 °C) |
| Solubility in butyrolactone | 12 g/100 ml (25 °C) 20 g/100 ml (60 °C) 35 g/100 ml (98 °C) |
| Solubility in cyclohexanone | 1 g/100 ml (25 °C) 8.9 g/100 ml (98 °C) |
| Solubility in dimethyl sulfoxide | 57 g/100 ml (25 °C) 68 g/100 ml (60 °C) 89 g/100 ml (98 °C)[3] |
| Vapor pressure | 2.41·10-8 mmHg at 25°C |
| Hazards | |
| Safety data sheet | Multihost IT |
| Related compounds | |
| Related compounds
|
RDX |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
HMX, also called octogen, is a powerful and relatively insensitive nitroamine high explosive, chemically related to RDX.
Like RDX, the compound's name is the subject of much speculation, having been variously listed as High Melting Explosive, Her Majesty's Explosive, High-velocity Military Explosive or High-Molecular-weight RDX.
Contents
Properties
Chemical
HMX will decompose if heated and can explode if a basting cap is used.
Physical
HMX is a white solid, odorless, practically insoluble in water and poorly soluble in most common organic solvents, but has good solubility in DMSO and butyrolactone.
DMF and NMP form a solvate with HMX.[4] A binary butyrolactone-DMSO solvent mixture is commonly used to dissolve HMX.[5]
Explosive
Because of its high molecular weight, HMX is one of the most potent chemical explosives manufactured commercially. It has a low shock and friction sensitivity, and cannot be detonated by igniting it, detonation requiring a blasting cap. HMX has a detonation velocity of 9,100 m/s and a R.E. factor of 1.7.
Availability
HMX, being a high explosive is not sold and can only be acquired by the military or research institutions.
Preparation
HMX can be prepared by nitrating hexamine using 70% nitric acid and ammonium nitrate in the presence of acetic anhydride and glacial acetic acid.[6][7]
One source indicates that 97% nitric acid also works best.[8]
Nitrating hexamine with concentrated nitric acid in the presence of ammonium nitrate, boron trifluoride, diethyl ether, acetic anhydride in acetic acid will also give HMX. The process takes around 75 min for completion at 44 °C, and gives an yield of around 16-17%.[9]
Projects
- Make blasting charges
Handling
Safety
At present, the information needed to determine if HMX causes cancer is insufficient. Due to the lack of information, EPA has determined that HMX is not classifiable as to its human carcinogenicity.
The available data on the effects on human health of exposure to HMX are limited. HMX causes CNS effects similar to those of RDX, but at considerably higher doses.
Storage
HMX is best kept in plastic or wooden containers. Do not store it for long.
Disposal
Controlled incineration should suffice.
Biodegradation using microorganisms has been successfully tried.[10]
References
- ↑ Liang, Lixuan; Cao, Dan; Song, Jinhong; Huang, Haifeng; Wang, Kai; Bian, Chengming; Dong, Xiao; Zhou, Zhiming; Journal of Materials Chemistry A; vol. 1; nb. 31; (2013); p. 8857 - 8865
- ↑ Monteil-Rivera, Fanny; Groom, Carl; Hawari, Jalal; Environmental Science and Technology; vol. 37; nb. 17; (2003); p. 3878 - 3884
- ↑ https://apps.dtic.mil/dtic/tr/fulltext/u2/773078.pdf
- ↑ https://pubs.acs.org/doi/pdf/10.1021/je60064a032
- ↑ https://apps.dtic.mil/dtic/tr/fulltext/u2/773078.pdf
- ↑ https://patents.google.com/patent/US2983725A
- ↑ http://www.sciencemadness.org/talk/viewthread.php?tid=32296
- ↑ https://www.nrcresearchpress.com/doi/pdf/10.1139/v52-088
- ↑ Huang, Chi-Yu; Mah, Robert A.; Que Hee, Shane S.; Journal of Labelled Compounds and Radiopharmaceuticals; vol. 41; nb. 5; (1998); p. 377 - 385
- ↑ https://www.researchgate.net/publication/226822722_Biodegradation_of_high_explosive_production_effluent_containing_RDX_and_HMX_by_denitrifying_bacteria