Difference between revisions of "1,2-Dichloroethane"
| Line 19: | Line 19: | ||
*Make PVC | *Make PVC | ||
| − | ==Safety== | + | ==Handling== |
| + | ===Safety=== | ||
Ethylene dichloride is toxic (by inhalation), flammable, and a [[carcinogen]]. All these hazards are amplified by the chemical's volatile nature. This chemical is also unstable when in the presence of [[aluminium]], [[zinc]], and [[iron]]<ref>http://en.wikipedia.org/wiki/1,2-Dichloroethane</ref>. | Ethylene dichloride is toxic (by inhalation), flammable, and a [[carcinogen]]. All these hazards are amplified by the chemical's volatile nature. This chemical is also unstable when in the presence of [[aluminium]], [[zinc]], and [[iron]]<ref>http://en.wikipedia.org/wiki/1,2-Dichloroethane</ref>. | ||
| + | |||
| + | ===Storage=== | ||
| + | |||
| + | ===Disposal=== | ||
==References== | ==References== | ||
<references/> | <references/> | ||
===Relevant Sciencemadness threads=== | ===Relevant Sciencemadness threads=== | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=3537 1,2-Dichloroethane from Acetylene and HCl] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=24404 Properties of 1,2-dichloroethane, interesting experiments?] | ||
| + | |||
[[Category:Polar solvents]] | [[Category:Polar solvents]] | ||
[[Category:Organic compounds]] | [[Category:Organic compounds]] | ||
Revision as of 20:33, 21 November 2015
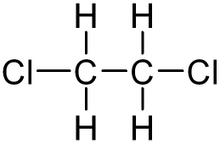
1,2-dichloroethane, or ethylene dichloride, is a common solvent and reagent, going by the abbreviations EDC and 1,2-DCA. It is also used to make vinyl chloride, the major precursor to polyvinyl chloride, commonly known as PVC.
Contents
Properties
Chemical
Ethylene dichloride is a slightly reactive polar solvent. It is sometimes used as a precursor to ethylenediamine and 1,1,1-trichloroethane[1].
Physical
Ethylene dichloride is colorless and has a high index of refraction, giving it a shiny appearance, similar to chloroform, which it also smells similarly to. It is a versatile solvent, though it does form azeotropes with water and many other solvents.
Availability
1,2-Dichloroethane is mostly bought from chemical suppliers rather than ordinary retail stores due to its inherent dangers.
Preparation
1,2-Dichloroethane can be prepared by bubbling chlorine through ethene solution (Eg. dissolved in Carbon Tetrachloride) with a Iron(III) chloride catalyst, or bubbling oxygen through a solution of 1 molar part ethene to four molar parts Hydrochloric acid with a Copper(II) chloride catalyst. The first procedure produces pure 1,2-dichloroethane, while the second results in water as well[2].
Projects
- Ethylenediamine synthesis
- Make PVC
Handling
Safety
Ethylene dichloride is toxic (by inhalation), flammable, and a carcinogen. All these hazards are amplified by the chemical's volatile nature. This chemical is also unstable when in the presence of aluminium, zinc, and iron[3].
Storage
Disposal
References
- ↑ http://en.wikipedia.org/wiki/1,2-Dichloroethane
- ↑ http://en.wikipedia.org/wiki/1,2-Dichloroethane
- ↑ http://en.wikipedia.org/wiki/1,2-Dichloroethane