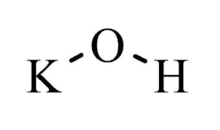Difference between revisions of "Potassium hydroxide"
| Line 136: | Line 136: | ||
This method can be performed by the amateur chemist, though it requires corrosion resistant containers when drying or concentrating the potassium hydroxide. | This method can be performed by the amateur chemist, though it requires corrosion resistant containers when drying or concentrating the potassium hydroxide. | ||
| − | Heating potassium carbonate at over 891 °C will yield potassium oxide that will quickly react with water to form hydroxide. It will also react with any carbon dioxide immediately at lower temperatures. This method is not very practical, as it requires | + | Heating potassium carbonate at over 891 °C will yield potassium oxide that will quickly react with water to form hydroxide. It will also react with any carbon dioxide immediately at lower temperatures. This method is not very practical, as it requires high temperatures. |
Electrolysis of a saturated solution of KCl with an ion exchange membrane will also yield potassium hydroxide. | Electrolysis of a saturated solution of KCl with an ion exchange membrane will also yield potassium hydroxide. | ||
| − | Another route, though expensive, involves the reaction of [[potassium | + | Another route, though expensive, involves the reaction of [[potassium]] metal or potassium oxide with water. |
==Projects== | ==Projects== | ||
Revision as of 21:55, 16 August 2015
| Names | |
|---|---|
| IUPAC name
Potassium hydroxide
| |
| Other names
Caustic potash
| |
| Properties | |
| KOH | |
| Molar mass | 56.106 |
| Appearance | White solid, deliquescent |
| Odor | Odorless |
| Melting point | 406 °C (763 °F; 679 K) |
| Boiling point | 1,327 °C (2,421 °F; 1,600 K) |
| 850 g/L (-23.2 °C) 970 g/L (0 °C) 1210 g/L (25 °C) 1383 g/L (50 °C) 1629 g/L (100 °C) | |
| Related compounds | |
| Related alkali hydroxides
|
lithium hydroxide sodium hydroxide rubidium hydroxide caesium hydroxide |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Potassium hydroxide is a white solid with the formula KOH. It finds many applications in the lab as a general base, though sodium hydroxide is generally preferred as it is easier to find and slightly cheaper.
Contents
Properties
Chemical
Potassium hydroxide dissolves in water and fully dissociates to give a solution of potassium ions and hydroxide ions. Potassium hydroxide solutions will very readily absorb atmospheric carbon dioxide to form potassium carbonate, and must be protected from the atmosphere. Being a strong base, potassium hydroxide also has the ability to dissolve glass (particularly so at higher temperatures) so solution must be kept in a chemically resistant plastic such as high density polyethylene (HDPE).
Potassium hydroxide can be reacted with the corresponding acids to yield potassium salts. Potassium hydroxide is used along with manganese dioxide and an oxidizer to form crude potassium manganate at very high temperatures.
Potassium hydroxide can also be used in a reaction with magnesium powder or shavings in an inert, high boiling solvent with a tertiary alcohol catalyst to yield clean spheres of potassium metal.
Physical
Potassium hydroxide is a waxy white solid which is extremely hygroscopic. It is very soluble in water and its dissolution is highly exothermic. One-hundred percent potassium hydroxide is very difficult to create due to the hygroscopic nature and commercial samples generally contain around 10% water.
Availability
Potassium hydroxide is available reasonably cheaply from soap making suppliers and biodiesel companies such as Duda Diesel. It is also available from Elemental Scientific at a slightly higher cost.
Certain barbecue cleaning products contain 25% KOH.
Preparation
Preparation of solid potassium hydroxide is difficult in an amateur setting due to the highly hygroscopic nature of potassium hydroxide.
Historically, potassium hydroxide was made by reacting potassium carbonate (potash) with calcium hydroxide (slaked lime).
- Ca(OH)2 + K2CO3 → CaCO3 + 2 KOH
This method can be performed by the amateur chemist, though it requires corrosion resistant containers when drying or concentrating the potassium hydroxide.
Heating potassium carbonate at over 891 °C will yield potassium oxide that will quickly react with water to form hydroxide. It will also react with any carbon dioxide immediately at lower temperatures. This method is not very practical, as it requires high temperatures.
Electrolysis of a saturated solution of KCl with an ion exchange membrane will also yield potassium hydroxide.
Another route, though expensive, involves the reaction of potassium metal or potassium oxide with water.
Projects
- Make potassium permanganate solution
- Produce clean potassium metal
- Make liquid soap
- Make an alkali battery
- Identify certain species of fungi
Handling
Safety
Potassium hydroxide is a strong base and is therefore highly caustic, but is not toxic in the sense that it shuts down chemical processes, like arsenic or cyanide. The potassium ion itself is nontoxic when taken orally, but injected into the blood stream it becomes highly toxic and may cause cardiac arrest. The primary danger is saponification of tissue on contact with the hydroxide ions.
Storage
Potassium hydroxide should be stored in closed thick plastic containers, in a dry and cool place, away from any corrosive vapors. Avoid storing it in glass bottles.
Disposal
Potassium hydroxide will turn to potassium carbonate if left in open air, which poses no toxicity to the environment and it's even used as a source of potassium for plants.