Difference between revisions of "Sodium carbonate"
(→Chemical) |
|||
| (7 intermediate revisions by the same user not shown) | |||
| Line 1: | Line 1: | ||
| − | + | {{Chembox | |
| − | [[ | + | | Name = Sodium carbonate |
| − | '''Sodium carbonate''', sometimes called '''washing soda '''or '''soda ash''', is an inorganic weak [[base]] with chemical formula Na<sub>2</sub>CO<sub>3</sub>. One of few water-soluble carbonates, sodium carbonate finds abundant use as a cheap source of the [[carbonate | + | | Reference = |
| + | | IUPACName = Sodium carbonate | ||
| + | | PIN = | ||
| + | | SystematicName = | ||
| + | | OtherNames = Disodium carbonate<br>Natrite<br>Soda ash<br>Washing soda | ||
| + | <!-- Images --> | ||
| + | | ImageFile = | ||
| + | | ImageSize = | ||
| + | | ImageAlt = | ||
| + | | ImageName = | ||
| + | | ImageCaption = | ||
| + | | ImageFile1 = Sodium carbonate anhydrous by Zts16.jpg | ||
| + | | ImageSize1 = 250 | ||
| + | | ImageAlt1 = | ||
| + | | ImageName1 = | ||
| + | | ImageCaption1 = Anhydrous sodium carbonate | ||
| + | | ImageFile2 = Sodium carbonate Wiki.png | ||
| + | | ImageSize2 = 200 | ||
| + | | ImageAlt2 = | ||
| + | | ImageName2 = | ||
| + | | ImageCaption2 = | ||
| + | | ImageFile3 = | ||
| + | | ImageSize3 = | ||
| + | | ImageAlt3 = | ||
| + | | ImageName3 = | ||
| + | | ImageFileL1 = | ||
| + | | ImageSizeL1 = | ||
| + | | ImageAltL1 = | ||
| + | | ImageNameL1 = | ||
| + | | ImageFileR1 = | ||
| + | | ImageSizeR1 = | ||
| + | | ImageAltR1 = | ||
| + | | ImageNameR1 = | ||
| + | | ImageFileL2 = | ||
| + | | ImageSizeL2 = | ||
| + | | ImageAltL2 = | ||
| + | | ImageNameL2 = | ||
| + | | ImageFileR2 = | ||
| + | | ImageSizeR2 = | ||
| + | | ImageAltR2 = | ||
| + | | ImageNameR2 = | ||
| + | <!-- Sections --> | ||
| + | | Section1 = {{Chembox Identifiers | ||
| + | | 3DMet = | ||
| + | | Abbreviations = | ||
| + | | SMILES = | ||
| + | }} | ||
| + | | Section2 = {{Chembox Properties | ||
| + | | AtmosphericOHRateConstant = | ||
| + | | Appearance = White solid | ||
| + | | BoilingPt = | ||
| + | | BoilingPtC = | ||
| + | | BoilingPt_ref = | ||
| + | | BoilingPt_notes = Decomposes | ||
| + | | Density = 2.54 g/cm<sup>3</sup> (25 °C, anhydrous)<br> 1.92 g/cm<sup>3</sup> (856 °C)<br> 2.25 g/cm<sup>3</sup> (monohydrate)<br> 1.51 g/cm<sup>3</sup> (heptahydrate)<br> 1.46 g/cm<sup>3</sup> (decahydrate) | ||
| + | | Formula = Na<sub>2</sub>CO<sub>3</sub> | ||
| + | | HenryConstant = | ||
| + | | LogP = | ||
| + | | MolarMass = 105.9888 g/mol (anhydrous)<br>286.1416 g/mol (decahydrate) | ||
| + | | MeltingPt = | ||
| + | | MeltingPtC = 851 | ||
| + | | MeltingPt_ref = | ||
| + | | MeltingPt_notes = <br> decomposes (anhydrous)<br> {{convert|100|C|F K}}<br> decomposes (monohydrate)<br> {{convert|33.5|C|F K}}<br> decomposes (heptahydrate)<br> {{convert|34|C|F K}}<br> (decahydrate) | ||
| + | | Odor = Odorless | ||
| + | | pKa = | ||
| + | | pKb = 3.67 | ||
| + | | Solubility = ''Monohydrate:''<br> 50.31 g/100 ml (29.9 °C)<br> 48.1 g/100 ml (41.9 °C)<br> 45.62 g/100 ml (60 °C)<br> 43.6 g/100 ml (100 °C)<hr>''Heptahydrate:''<br> 48.69 g/100 ml (34.8 °C)<hr>''Decahydrate:''<br>7 g/100 ml (0 °C)<br> 16.4 g/100 ml (15 °C)<br> 34.07 g/100 ml (27.8 °C)<br> | ||
| + | | SolubleOther = Soluble in aq. alkali, [[glycerol]]<br> Slightly soluble in aq. [[alcohol]], [[ethylene glycol]]<br>Insoluble in [[acetone]], liq. [[ammonia]], [[benzonitrile]], [[carbon disulfide|CS<sub>2</sub>]], [[ethanol]], [[ethyl acetate]] | ||
| + | | Solubility1 = 98.3 g/100 g (15.5 °C) | ||
| + | | Solvent1 = glycerol | ||
| + | | Solubility2 = 3.46 g/100 g (20 °C) | ||
| + | | Solvent2 = ethylene glycol | ||
| + | | Solubility3 = 0.05 g/100 g | ||
| + | | Solvent3 = dimethylformamide | ||
| + | | VaporPressure = ~0 mmHg | ||
| + | }} | ||
| + | | Section3 = {{Chembox Structure | ||
| + | | Coordination = Octahedral (Na<sup>+</sup>, anhydrous) | ||
| + | | CrystalStruct = Monoclinic (γ-form, β-form, δ-form, anhydrous)<br>Orthorhombic (monohydrate, heptahydrate) | ||
| + | | MolShape = | ||
| + | }} | ||
| + | | Section4 = {{Chembox Thermochemistry | ||
| + | | DeltaGf = −1,044.4 kJ/mol | ||
| + | | DeltaHc = | ||
| + | | DeltaHf = −1,130.7 kJ/mol | ||
| + | | Entropy = 135 J·mol<sup>-1</sup>·K<sup>-1</sup> | ||
| + | | HeatCapacity = 112.3 J·mol<sup>-1</sup>·K<sup>-1</sup> | ||
| + | }} | ||
| + | | Section5 = {{Chembox Explosive | ||
| + | | ShockSens = | ||
| + | | FrictionSens = | ||
| + | | DetonationV = | ||
| + | | REFactor = | ||
| + | }} | ||
| + | | Section6 = {{Chembox Hazards | ||
| + | | AutoignitionPt = Non-flammable | ||
| + | | ExploLimits = Non-explosive | ||
| + | | ExternalMSDS = [https://www.docdroid.net/uAqx265/sodium-carbonate-anhydrous-sa.pdf.html Sigma-Aldrich] (anhydrous)<br>[https://www.docdroid.net/VCWQZx7/sodium-carbonate-decahydrate-sa.pdf.html Sigma-Aldrich] (decahydrate) | ||
| + | | FlashPt = Non-flammable | ||
| + | | LD50 = 4,090 mg/kg (rat, oral) | ||
| + | | LC50 = | ||
| + | | MainHazards = Irritant | ||
| + | | NFPA-F = | ||
| + | | NFPA-H = | ||
| + | | NFPA-R = | ||
| + | | NFPA-S = | ||
| + | }} | ||
| + | | Section7 = {{Chembox Related | ||
| + | | OtherAnions = | ||
| + | | OtherCations = | ||
| + | | OtherFunction = | ||
| + | | OtherFunction_label = | ||
| + | | OtherCompounds = [[Lithium carbonate]]<br>[[Potassium carbonate]] | ||
| + | }} | ||
| + | }} | ||
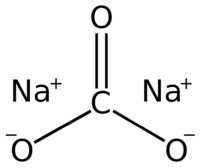
| + | '''Sodium carbonate''', sometimes called '''washing soda''' or '''soda ash''', is an inorganic weak [[base]] with chemical formula '''Na<sub>2</sub>CO<sub>3</sub>'''. One of few water-soluble carbonates, sodium carbonate finds abundant use as a cheap source of the [[carbonate]] ion, and also as a precursor to other sodium salts. Sodium carbonate occurs in nature as mineral '''Natrite'''. | ||
==Properties== | ==Properties== | ||
===Chemical=== | ===Chemical=== | ||
Sodium carbonate will react with acids to form sodium salts and carbon dioxide. | Sodium carbonate will react with acids to form sodium salts and carbon dioxide. | ||
| + | |||
| + | : Na<sub>2</sub>CO<sub>3</sub> + 2 HCl → 2 NaCl + H<sub>2</sub>O + CO<sub>2</sub> | ||
| + | : Na<sub>2</sub>CO<sub>3</sub> + 2 HCOOH → 2 HCOONa + H<sub>2</sub>O + CO<sub>2</sub> | ||
At very high temperatures, it decomposes to form sodium oxide and carbon dioxide. | At very high temperatures, it decomposes to form sodium oxide and carbon dioxide. | ||
| + | |||
| + | : Na<sub>2</sub>CO<sub>3</sub> → Na<sub>2</sub>O + CO<sub>2</sub> | ||
| + | |||
| + | However, unlike other carbonates, the decomposition of alkali carbonates is complicated. | ||
| + | |||
| + | Bubbling carbon dioxide in an aqueous solution of sodium carbonate will give [[sodium bicarbonate]]. | ||
| + | |||
| + | : Na<sub>2</sub>CO<sub>3</sub> + H<sub>2</sub>O + CO<sub>2</sub> → 2 NaHCO<sub>3</sub> | ||
| + | |||
| + | If [[sulfur dioxide]] is used, [[sodium sulfite]] is produced. Excess SO<sub>2</sub> gives [[sodium bisulfite]]. | ||
| + | |||
| + | : Na<sub>2</sub>CO<sub>3</sub> + H<sub>2</sub>O + SO<sub>2</sub> → Na<sub>2</sub>SO<sub>3</sub> + H<sub>2</sub>O + CO<sub>2</sub> | ||
| + | : Na<sub>2</sub>CO<sub>3</sub> + H<sub>2</sub>O + 2 SO<sub>2</sub> → 2 NaHSO<sub>3</sub> + CO<sub>2</sub> | ||
| + | |||
| + | This route can be used to remove the sulfur dioxide produced from various reactions. | ||
Sodium carbonate is often used in ore refining, in which it is fused with metal oxides at high temperatures to generate water-soluble compounds of the metal. An example of this is the roasting of [[chromium(III) oxide]] with sodium carbonate in the presence of oxygen to generate [[sodium dichromate]]. | Sodium carbonate is often used in ore refining, in which it is fused with metal oxides at high temperatures to generate water-soluble compounds of the metal. An example of this is the roasting of [[chromium(III) oxide]] with sodium carbonate in the presence of oxygen to generate [[sodium dichromate]]. | ||
| + | |||
| + | : Na<sub>2</sub>CO<sub>3</sub> + Cr<sub>2</sub>O<sub>3</sub> + O<sub>2</sub> → Na<sub>2</sub>Cr<sub>2</sub>O<sub>7</sub> + CO<sub>2</sub> | ||
===Physical=== | ===Physical=== | ||
| − | + | Sodium carbonate is a white hydgoscopic compound, soluble in water but insoluble in organic solvents. It reacts with acids, sulfur and nitrogen oxides, as well as halogens, the reaction proceeding faster in the presence of water. Sodium carbonate crystallizes from water to form three different hydrates: monohydrate, heptahydrate and decahydrate. | |
| − | Sodium carbonate is a white hydgoscopic compound, soluble in water but insoluble in organic solvents. Sodium carbonate crystallizes from water to form three different hydrates: monohydrate, heptahydrate and decahydrate. | + | |
==Preparation and acquisition== | ==Preparation and acquisition== | ||
Historically, sodium carbonate could be produced by leaching the ashes of burnt seaweed or glassworth, but this leads to an impure product. Today, sodium carbonate can be easily and cheaply prepared by the thermal decomposition of store-bought [[sodium bicarbonate]] (baking soda), which begins at 50 °C, but is more rapidly and completely accomplished at temperatures over 200 °C. Alternatively, it can be be purchased as washing soda or pH-increasing formulas in grocery or department stores, both of which are sometimes as cheap as $1/lb, or as soda ash, which is used as a mordant for textile dying. | Historically, sodium carbonate could be produced by leaching the ashes of burnt seaweed or glassworth, but this leads to an impure product. Today, sodium carbonate can be easily and cheaply prepared by the thermal decomposition of store-bought [[sodium bicarbonate]] (baking soda), which begins at 50 °C, but is more rapidly and completely accomplished at temperatures over 200 °C. Alternatively, it can be be purchased as washing soda or pH-increasing formulas in grocery or department stores, both of which are sometimes as cheap as $1/lb, or as soda ash, which is used as a mordant for textile dying. | ||
| + | |||
| + | Another convenient route is to add [[sodium hydroxide]] to [[ammonium carbonate]] or [[ammonium bicarbonate|bicarbonate]], then boil the solution until it dries completely. This produces sodium carbonate, while ammonia is produced as side-product. This route is attractive if you have old or contaminated sodium hydroxide, which you need to dispose of. | ||
| + | |||
| + | Lastly, leaving sodium hydroxide solution in open air will slowly form sodium carbonate. Make sure to do this in an area with clean air, as polluted air also contains sulfur and nitrogen oxides which will contaminate your sodium carbonate with sodium sulfate and sodium nitrate. Another problem, is that the resulting sodium carbonate will "grow" on the sides of your container and may even completely cover your container if you leave it long enough. Make sure to periodically check the evolution of the sodium carbonate. | ||
==Uses== | ==Uses== | ||
| − | As a basic sodium compound, the addition of most inorganic and some organic acids yields a sodium salt of that acid, and due to the almost universal solubility of sodium compounds, these salts can be used in preparing other metal salts of the same anion(for example, the preparation of the lesser-known [[copper aspirinate]] using [[sodium aspirinate]] and a soluble copper salt). Note that neutralizing a carbonate with an acid evolves large amounts of [[carbon dioxide]] and causes lots of bubbling. For this reason, [[Sodium hydroxide]] is most often used for [[titration]]. | + | As a basic sodium compound, the addition of most inorganic and some organic acids yields a sodium salt of that acid, and due to the almost universal solubility of sodium compounds, these salts can be used in preparing other metal salts of the same anion(for example, the preparation of the lesser-known [[Copper(II) acetylsalicylate|copper aspirinate]] using [[Sodium acetylsalicylate|sodium aspirinate]] and a soluble copper salt). Note that neutralizing a carbonate with an acid evolves large amounts of [[carbon dioxide]] and causes lots of bubbling. For this reason, [[Sodium hydroxide]] is most often used for [[titration]]. |
| − | Because many insoluble [[transition metal]] [[carbonate]]s are used as precursors to salts of those metals, sodium carbonate is an invaluable reagent in forming a large number of compounds, first through precipitation of the insoluble carbonate in a [[double replacement | + | Because many insoluble [[transition metal]] [[carbonate]]s are used as precursors to salts of those metals, sodium carbonate is an invaluable reagent in forming a large number of compounds, first through precipitation of the insoluble carbonate in a [[double replacement]] reaction. |
Bubbling [[carbon dioxide]] through a sodium carbonate solution yields [[sodium bicarbonate]]. | Bubbling [[carbon dioxide]] through a sodium carbonate solution yields [[sodium bicarbonate]]. | ||
| Line 33: | Line 171: | ||
===Disposal=== | ===Disposal=== | ||
| − | Sodium carbonate can be safely poured down the drain. | + | Sodium carbonate can be safely poured down the drain or dumped in trash. Avoid dumping it in the ground, as excess sodium is harmful to plants. |
| + | |||
| + | ==Gallery== | ||
| + | <gallery widths="200" position="center" columns="4" orientation="none"> | ||
| + | Sodium carbonate decahydrate.jpg|Sodium carbonate decahydrate crystals | ||
| + | </gallery> | ||
==References== | ==References== | ||
| Line 39: | Line 182: | ||
===Relevant Sciencemadness threads=== | ===Relevant Sciencemadness threads=== | ||
*[http://www.sciencemadness.org/talk/viewthread.php?tid=21167 sodium carbonate - wikipedia - what?] | *[http://www.sciencemadness.org/talk/viewthread.php?tid=21167 sodium carbonate - wikipedia - what?] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=71493 Sodium Carbonate and Aluminum reaction] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=7075 making sodium carbonate] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=1243 sodium carbonate: where do i buy it?] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=2604 Sodium Carbonate] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=612 Sodium carbonate?] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=27271 Dehydrating sodium carbonate] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=5643 equation to make sodium carbonate from acetone and sodium hydroxide] | ||
| + | *[http://www.sciencemadness.org/talk/viewthread.php?tid=73040 Electrolysis of sodium carbonate with copper electrodes ] | ||
[[Category:Chemical compounds]] | [[Category:Chemical compounds]] | ||
| Line 44: | Line 195: | ||
[[Category:Sodium compounds]] | [[Category:Sodium compounds]] | ||
[[Category:Carbonates]] | [[Category:Carbonates]] | ||
| − | |||
[[Category:Bases]] | [[Category:Bases]] | ||
[[Category:Hygroscopic compounds]] | [[Category:Hygroscopic compounds]] | ||
| Line 50: | Line 200: | ||
[[Category:Readily available chemicals]] | [[Category:Readily available chemicals]] | ||
[[Category:Minerals]] | [[Category:Minerals]] | ||
| + | [[Category:Essential reagents]] | ||
Latest revision as of 14:39, 18 November 2023
 Anhydrous sodium carbonate
| |
| |
| Names | |
|---|---|
| IUPAC name
Sodium carbonate
| |
| Other names
Disodium carbonate
Natrite Soda ash Washing soda | |
| Properties | |
| Na2CO3 | |
| Molar mass | 105.9888 g/mol (anhydrous) 286.1416 g/mol (decahydrate) |
| Appearance | White solid |
| Odor | Odorless |
| Density | 2.54 g/cm3 (25 °C, anhydrous) 1.92 g/cm3 (856 °C) 2.25 g/cm3 (monohydrate) 1.51 g/cm3 (heptahydrate) 1.46 g/cm3 (decahydrate) |
| Melting point | 851 °C (1,564 °F; 1,124 K) decomposes (anhydrous) 100 °C (212 °F; 373 K) decomposes (monohydrate) 33.5 °C (92.3 °F; 306.6 K) decomposes (heptahydrate) 34 °C (93 °F; 307 K) (decahydrate) |
| Boiling point | Decomposes |
| Monohydrate: 50.31 g/100 ml (29.9 °C) 48.1 g/100 ml (41.9 °C) 45.62 g/100 ml (60 °C) 43.6 g/100 ml (100 °C) Heptahydrate: 48.69 g/100 ml (34.8 °C) Decahydrate: 7 g/100 ml (0 °C) 16.4 g/100 ml (15 °C) 34.07 g/100 ml (27.8 °C) | |
| Solubility | Soluble in aq. alkali, glycerol Slightly soluble in aq. alcohol, ethylene glycol Insoluble in acetone, liq. ammonia, benzonitrile, CS2, ethanol, ethyl acetate |
| Solubility in glycerol | 98.3 g/100 g (15.5 °C) |
| Solubility in ethylene glycol | 3.46 g/100 g (20 °C) |
| Solubility in dimethylformamide | 0.05 g/100 g |
| Vapor pressure | ~0 mmHg |
| Thermochemistry | |
| Std molar
entropy (S |
135 J·mol-1·K-1 |
| Std enthalpy of
formation (ΔfH |
−1,130.7 kJ/mol |
| Hazards | |
| Safety data sheet | Sigma-Aldrich (anhydrous) Sigma-Aldrich (decahydrate) |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
| LD50 (Median dose)
|
4,090 mg/kg (rat, oral) |
| Related compounds | |
| Related compounds
|
Lithium carbonate Potassium carbonate |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Sodium carbonate, sometimes called washing soda or soda ash, is an inorganic weak base with chemical formula Na2CO3. One of few water-soluble carbonates, sodium carbonate finds abundant use as a cheap source of the carbonate ion, and also as a precursor to other sodium salts. Sodium carbonate occurs in nature as mineral Natrite.
Contents
Properties
Chemical
Sodium carbonate will react with acids to form sodium salts and carbon dioxide.
- Na2CO3 + 2 HCl → 2 NaCl + H2O + CO2
- Na2CO3 + 2 HCOOH → 2 HCOONa + H2O + CO2
At very high temperatures, it decomposes to form sodium oxide and carbon dioxide.
- Na2CO3 → Na2O + CO2
However, unlike other carbonates, the decomposition of alkali carbonates is complicated.
Bubbling carbon dioxide in an aqueous solution of sodium carbonate will give sodium bicarbonate.
- Na2CO3 + H2O + CO2 → 2 NaHCO3
If sulfur dioxide is used, sodium sulfite is produced. Excess SO2 gives sodium bisulfite.
- Na2CO3 + H2O + SO2 → Na2SO3 + H2O + CO2
- Na2CO3 + H2O + 2 SO2 → 2 NaHSO3 + CO2
This route can be used to remove the sulfur dioxide produced from various reactions.
Sodium carbonate is often used in ore refining, in which it is fused with metal oxides at high temperatures to generate water-soluble compounds of the metal. An example of this is the roasting of chromium(III) oxide with sodium carbonate in the presence of oxygen to generate sodium dichromate.
- Na2CO3 + Cr2O3 + O2 → Na2Cr2O7 + CO2
Physical
Sodium carbonate is a white hydgoscopic compound, soluble in water but insoluble in organic solvents. It reacts with acids, sulfur and nitrogen oxides, as well as halogens, the reaction proceeding faster in the presence of water. Sodium carbonate crystallizes from water to form three different hydrates: monohydrate, heptahydrate and decahydrate.
Preparation and acquisition
Historically, sodium carbonate could be produced by leaching the ashes of burnt seaweed or glassworth, but this leads to an impure product. Today, sodium carbonate can be easily and cheaply prepared by the thermal decomposition of store-bought sodium bicarbonate (baking soda), which begins at 50 °C, but is more rapidly and completely accomplished at temperatures over 200 °C. Alternatively, it can be be purchased as washing soda or pH-increasing formulas in grocery or department stores, both of which are sometimes as cheap as $1/lb, or as soda ash, which is used as a mordant for textile dying.
Another convenient route is to add sodium hydroxide to ammonium carbonate or bicarbonate, then boil the solution until it dries completely. This produces sodium carbonate, while ammonia is produced as side-product. This route is attractive if you have old or contaminated sodium hydroxide, which you need to dispose of.
Lastly, leaving sodium hydroxide solution in open air will slowly form sodium carbonate. Make sure to do this in an area with clean air, as polluted air also contains sulfur and nitrogen oxides which will contaminate your sodium carbonate with sodium sulfate and sodium nitrate. Another problem, is that the resulting sodium carbonate will "grow" on the sides of your container and may even completely cover your container if you leave it long enough. Make sure to periodically check the evolution of the sodium carbonate.
Uses
As a basic sodium compound, the addition of most inorganic and some organic acids yields a sodium salt of that acid, and due to the almost universal solubility of sodium compounds, these salts can be used in preparing other metal salts of the same anion(for example, the preparation of the lesser-known copper aspirinate using sodium aspirinate and a soluble copper salt). Note that neutralizing a carbonate with an acid evolves large amounts of carbon dioxide and causes lots of bubbling. For this reason, Sodium hydroxide is most often used for titration.
Because many insoluble transition metal carbonates are used as precursors to salts of those metals, sodium carbonate is an invaluable reagent in forming a large number of compounds, first through precipitation of the insoluble carbonate in a double replacement reaction.
Bubbling carbon dioxide through a sodium carbonate solution yields sodium bicarbonate.
Handling
Safety
Sodium carbonate is non-toxic, though due to its desiccant properties it may irritate the skin.
Storage
Sodium carbonate should be stored in closed bottles, away from moisture and acidic vapors.
Disposal
Sodium carbonate can be safely poured down the drain or dumped in trash. Avoid dumping it in the ground, as excess sodium is harmful to plants.
Gallery
References
Relevant Sciencemadness threads
- sodium carbonate - wikipedia - what?
- Sodium Carbonate and Aluminum reaction
- making sodium carbonate
- sodium carbonate: where do i buy it?
- Sodium Carbonate
- Sodium carbonate?
- Dehydrating sodium carbonate
- equation to make sodium carbonate from acetone and sodium hydroxide
- Electrolysis of sodium carbonate with copper electrodes
