| Pages:
1
2
3
4
5 |
clearly_not_atara
International Hazard
    
Posts: 2834
Registered: 3-11-2013
Member Is Offline
Mood: Big
|
|
Recently Loptr posted about the catalytic tosylation of alcohols with CoCl2:
https://www.sciencemadness.org/whisper/viewthread.php?tid=91...
However, a careful reading of the paper he attached shows only tosylations of secondary alcohols, and indeed, the paper claims that the reaction
prefers to tosylate secondary rather than primary alcohols. But the paper references an earlier paper, using Fe3+ on montmorillonite as a
heterogeneous catalyst for tosylation of primary alcohols with TsOH.
That paper, "Montmorillonite Clay Catalyzed Tosylation of Alcohols and Selective Monotosylation of Diols with p-Toluenesulfonic Acid: An Enviro-Economic
Route", is attached. In fact, this paper claims the preparation of methyl tosylate in 82% yield:
MeOH + TsOH [ClCH2CH2Cl, Fe3+/montmorillonite clay, reflux, 3 hours] >> MeOTs + H2O
When I discussed the CoCl2 paper on another forum, there were some concerns that the results might not replicate. The following points were raised:
- TsO- is a good leaving group
- The researchers are from India
- The paper was published in Tetrahedron Letters
In response to the first point, I want to point out that the reaction of triflic acid and alcohols produces some yield of alkyl triflates with no
catalyst:
https://en.wikipedia.org/wiki/Triflic_acid#Organic_chemistry
We know catalysts cannot change an equilibrium, only cause it to be reached, so the role of the catalyst is irrelevant when considering the reaction
energetics. The equilibrium:
MeOH2+ + TsO- >> << TsOMe + H2O
does not at first seem to lean right, but if you complete the equation:
MeOH2+ + TsO- + TsOH >> << TsOMe + H2O + TsOH >> << TsOMe + TsO- + H3O+
you can then understand why TsOMe is produced. H3O+ (pKa -1.6) is a weaker acid than MeOH2+ (pKa -2.5), so the reaction is actually driven forward by
the basicity of water, or alternatively, by the affinity of TsOH for H2O. It is therefore clear that using an excess of toluenesulfonic acid should
help drive the reaction to completion.
In response to "the researchers are from India", this paper actually comes from two very distinguished researchers who both have h-indexes over 50:
https://scholar.google.com/citations?user=R1MxH_EAAAAJ&h...
https://scholar.google.com/citations?user=XgHcVy0AAAAJ&h...
The ICT Mumbai where Prof. Kantam works is one of the top 20 universities in India. So I think these guys are better than your standard-issue Indian
researchers.
As for TetLet, I can't defend it, but the paper got cited 56 times without any complaints, which is a good sign. Overall, the method definitely
warrants further investigation, particularly since montmorillonite and aqueous FeCl3 are OTC. Amazon reviews laud the effectiveness of montmorillonite
for "detoxification from chemtrails", so as usual, crazy people keep us in business.
Intriguingly, the paper also claims the preparation of 1-tosyloxy-2-propanol from propylene glycol by the same method.
The preparation of the catalyst is as follows:
"K10 montmorillonite purchased from Fluka was used as
such. The chemical composition of this starting material
(main elements) is SiO2, 67.6; Al2O3, 14.6; Fe2O3, 2.9;
MgO, 1.8. Metal-exchanged montmorillonite:22 to 1 L of
1 M aqueous metal chloride solution, 80 g of K10 montmorillonite was added. Stirring was maintained for
16–30 h in order to saturate the exchange capacity of K10
montmorillonite. The clay suspension was centrifuged and
the supernatant solution was discarded. The clay catalyst
was washed each time with fresh distilled water until free
of chloride ions as indicated by AgNO3 test. The catalyst
was dried overnight in an oven at 1208C and finely ground in
a mortar."
Attachment: choudary2000.pdf (161kB)
This file has been downloaded 626 times
[Edited on 22-9-2018 by clearly_not_atara]
|
|
|
AvBaeyer
National Hazard
   
Posts: 655
Registered: 25-2-2014
Location: CA
Member Is Offline
Mood: No Mood
|
|
clearly-not-atara:
You obviously did not read the paper very carefully. The paper reports high yields of primary benzylic and allylic alcohols and even a modest yield of
a normal aliphatic primary alcohol which is very clearly shown in Table 2. How could you miss that? Indeed secondary aliphatic alcohols appear to be
selected for over primary ones but not to complete exclusion. Please try to be a bit more thorough.
AvB
|
|
|
clearly_not_atara
International Hazard
    
Posts: 2834
Registered: 3-11-2013
Member Is Offline
Mood: Big
|
|
Quote: Originally posted by AvBaeyer  | clearly-not-atara:
You obviously did not read the paper very carefully. The paper reports high yields of primary benzylic and allylic alcohols and even a modest yield of
a normal aliphatic primary alcohol which is very clearly shown in Table 2. How could you miss that? Indeed secondary aliphatic alcohols appear to be
selected for over primary ones but not to complete exclusion. Please try to be a bit more thorough.
AvB |
My bad, I missed entry 5 in Table 2. I wouldn't normally count allylic and benzylic alcohols as
analogous to methanol.
But really my point in posting was to argue that these reactions are plausible despite the fact that the papers are from Indian researchers and
published in Tetrahedron Letters. I was originally going to write the same post defending the first paper, but in the other paper methyl
tosylate was actually synthesized, so of course it seemed more appropriate here.
People didn't seem as impressed by Loptr's post as I thought they would be, because discussions of methylating agents usually end with no good
answers. I thought maybe nobody was interested in the reaction because they didn't think it would work, but after thinking about it and reading other
related literature, I wanted to argue that it will work.
|
|
|
Loptr
International Hazard
    
Posts: 1348
Registered: 20-5-2014
Location: USA
Member Is Offline
Mood: Grateful
|
|
I started looking for more articles discussing catalytic tosylation and discovered this has been discussed before The Hive by psyloxy. I am not sure
what ever came of it.
https://the-hive.archive.erowid.org/forum/showflat/Cat-/Numb...
https://chemistry.mdma.ch/hiveboard/chemistrydiscourse/00050...
Another Indian/Tetrahedron paper, except this time using silicon tetrachloride. There is mention of reaction with MeOH getting an 87% yield.
An efficient and selective tosylation of alcohols with p-toluenesulfonic acid
https://www.sciencedirect.com/science/article/pii/S004040390...
Attachment: das2004.pdf (250kB)
This file has been downloaded 974 times
Another one that I have found uses ZrCl4 as catalyst.
Greene's Protective Groups in Organic Synthesis also mentions these three methods. YMMV.
[Edited on 24-9-2018 by Loptr]

"Question everything generally thought to be obvious." - Dieter Rams
|
|
|
Loptr
International Hazard
    
Posts: 1348
Registered: 20-5-2014
Location: USA
Member Is Offline
Mood: Grateful
|
|
Reaction of Carboxylic Acid Esters with p-Toluenesulfonic Acid

Attachment: nitta1985.pdf (742kB)
This file has been downloaded 594 times
"Question everything generally thought to be obvious." - Dieter Rams
|
|
|
clearly_not_atara
International Hazard
    
Posts: 2834
Registered: 3-11-2013
Member Is Offline
Mood: Big
|
|
^now that is awesome!
The paper made me wonder if methyl carbamate (bp 177 C) could be used instead of dimethyl carbonate (bp 90 C) since the latter's low boiling point was
blamed for a low yield. In both cases, the acid decomposes upon cleavage:
MeO2CNH2 + 2 TsOH >> MeOTs + CO2 + NH4OTs
The amine should not be alkylated as long as the reaction mixture is not basified in situ -- ammonium tosylate can be washed away with cold water,
which should not dissolve MeOTs.
It's my understanding that methyl carbamate is actually produced more easily from methanol + urea than DMC. The first NH2 comes off more easily than
the second. Methyl carbamate might also be produced from the reaction MeOH + NaOCN + acid, as long as that reaction can be performed safely --
isocyanic acid is a little scary, but NaOCN is very easy to make.
|
|
|
Loptr
International Hazard
    
Posts: 1348
Registered: 20-5-2014
Location: USA
Member Is Offline
Mood: Grateful
|
|
Methyl tosylate seems to be a very useful reagent. I am finding a treasure trove of interesting reactions.
Nickel-Catalyzed Reductive Methylation of Alkyl Halides and Acid Chlorides with Methyl p-Tosylate
https://pubs.acs.org/doi/10.1021/ol502682q
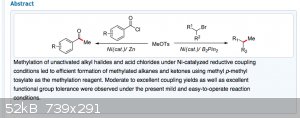
Attachment: Nickel-Catalyzed Reductive Methylation of Alkyl Halides and Acid Chlorides with Methyl p-Tosylate.pdf (479kB)
This file has been downloaded 596 times
[Edited on 25-9-2018 by Loptr]
"Question everything generally thought to be obvious." - Dieter Rams
|
|
|
byko3y
National Hazard
   
Posts: 721
Registered: 16-3-2015
Member Is Offline
Mood: dooM
|
|
That's a modification of reaction known for at least thirty years:
https://doi.org/10.1016/S0040-4039(00)81952-8 - Metallic nickel: A coupling reagent of benzyl halides and acyl halides to yield benzyl ketones
https://dx.doi.org/10.1021/jo00209a006 - Metallic nickel-mediated synthesis of ketones by the reaction of benzylic, allylic, vinylic, and
pentafluorophenyl halides with acid halides
|
|
|
monolithic
Hazard to Others
  
Posts: 436
Registered: 5-3-2018
Member Is Offline
Mood: No Mood
|
|
Are these reagents any more "OTC" or safer than dimethyl carbonate or trimethyl phosphate?
|
|
|
clearly_not_atara
International Hazard
    
Posts: 2834
Registered: 3-11-2013
Member Is Offline
Mood: Big
|
|
Everything in this thread is more OTC than trimethyl phosphate. Given an arbitrary compound containing "methyl", it's probably more OTC than methyl
phosphate. (MeO)3PO is made exclusively from the reaction of alcohols with phosphoryl chloride, which is highly restricted because it's an LSD
precursor.
The OTC synthesis of dimethyl carbonate is believed to be fully achievable, but AFAICT it has never been performed. Dimethyl carbonate also requires
pressure for methylation, so while it is safe to handle, it is less safe and not convenient to use.
In general methylating agents are not sold to the public, for obvious reasons. I would say that these direct esterifications of toluenesulfonic acid
are the most promising development I've ever seen in methylation reactions in amateur chemistry -- although, to be fair, nobody has tried it yet.
Methyl tosylate is certain to be highly carcinogenic if ingested, but it has a very low vapor pressure, so most of us believe it is much safer than
other strong methylating agents. Methyl phosphate and carbonate are much less reactive and therefore safer, although (MeO)3PO is probably a
carcinogen.
|
|
|
monolithic
Hazard to Others
  
Posts: 436
Registered: 5-3-2018
Member Is Offline
Mood: No Mood
|
|
I think dimethyl carbonate is being phased in as a VOC compliant solvent, so it is OTC, in a way. You can purchase a liter of it on Amazon right now.
|
|
|
Loptr
International Hazard
    
Posts: 1348
Registered: 20-5-2014
Location: USA
Member Is Offline
Mood: Grateful
|
|
Quote: Originally posted by monolithic  | | I think dimethyl carbonate is being phased in as a VOC compliant solvent, so it is OTC, in a way. You can purchase a liter of it on Amazon right now.
|
I am still confused why you said trimethyl phosphate was OTC. You can't make the phosphate esters from phosphoric acid.
"Question everything generally thought to be obvious." - Dieter Rams
|
|
|
clearly_not_atara
International Hazard
    
Posts: 2834
Registered: 3-11-2013
Member Is Offline
Mood: Big
|
|
Quote: Originally posted by monolithic  | | I think dimethyl carbonate is being phased in as a VOC compliant solvent, so it is OTC, in a way. You can purchase a liter of it on Amazon right now.
|
That makes it an awful lot easier to use the methyl tosylate procedure Loptr just posted.
Although, being that DMC is volatile, organic, and degrades to methanol, which as we all know causes blindness, I'm curious why it's considered to be
VOC compliant? Is it really considered safer than acetone or something? I know it's a battery electrolyte, but I'm pretty sure that methyl ethyl
carbonate "MEC" is preferred due to some sort of weird electrical properties.
|
|
|
monolithic
Hazard to Others
  
Posts: 436
Registered: 5-3-2018
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by Loptr  | Quote: Originally posted by monolithic  | | I think dimethyl carbonate is being phased in as a VOC compliant solvent, so it is OTC, in a way. You can purchase a liter of it on Amazon right now.
|
I am still confused why you said trimethyl phosphate was OTC. You can't make the phosphate esters from phosphoric acid. |
I thought I remembered reading a "somewhat" straightforward preparation of trimethyl phosphate, but I'm clearly wrong.
|
|
|
Loptr
International Hazard
    
Posts: 1348
Registered: 20-5-2014
Location: USA
Member Is Offline
Mood: Grateful
|
|
Quote: Originally posted by monolithic  | Quote: Originally posted by Loptr  | Quote: Originally posted by monolithic  | | I think dimethyl carbonate is being phased in as a VOC compliant solvent, so it is OTC, in a way. You can purchase a liter of it on Amazon right now.
|
I am still confused why you said trimethyl phosphate was OTC. You can't make the phosphate esters from phosphoric acid. |
I thought I remembered reading a "somewhat" straightforward preparation of trimethyl phosphate, but I'm clearly wrong. |
I thought you were going to have an OTC source for it! What a let down! 
"Question everything generally thought to be obvious." - Dieter Rams
|
|
|
deep_dreamer
Harmless

Posts: 9
Registered: 19-4-2018
Member Is Offline
Mood: No Mood
|
|
It seems that trimethyl phosphate may also be produced by reacting P2O5 with MeOH, yield about 35%. I have originally found the procedure as a german
translation on LambdaSyn, but this seems to be a copy of the original writeup:
https://chemistry.mdma.ch/hiveboard/rhodium/me3po4.html
|
|
|
monolithic
Hazard to Others
  
Posts: 436
Registered: 5-3-2018
Member Is Offline
Mood: No Mood
|
|
I believe this is what I was thinking about, although P2O5 isn't very OTC. 
|
|
|
clearly_not_atara
International Hazard
    
Posts: 2834
Registered: 3-11-2013
Member Is Offline
Mood: Big
|
|
Apparently aluminium phosphate decomposes to P2O5? People in the old thread couldn't find a use for P2O5, but Magpie reported that the reaction was
noticeable at 1000 C:
http://www.sciencemadness.org/talk/viewthread.php?tid=12694
Aluminium metaphosphate has been described and it might decompose sooner:
https://www.americanelements.com/aluminum-metaphosphate-1377...
but I've no idea how to make it. Nickel sulfate is used in the sulfur-iodine process as a precursor to SO3, so Ni3(PO4)2 or a nickel polyphosphate
might also be a good choice.
|
|
|
zed
International Hazard
    
Posts: 2285
Registered: 6-9-2008
Location: Great State of Jefferson, City of Portland
Member Is Offline
Mood: Semi-repentant Sith Lord
|
|
P2O5?
Well, you probably can't buy it at Ace hardware, but some pyrotechnics suppliers might have it.
It isn't restricted (I don't think); it's just a fussy reagent that must be handled carefully.
Available on both Ebay and Amazon, at an outrageous price.....
But, cheaper elsewhere.
https://www.ebay.com/itm/Phosphorus-pentoxide-99-5-Analytica...
[Edited on 16-10-2018 by zed]
[Edited on 16-10-2018 by zed]
[Edited on 16-10-2018 by zed]
|
|
|
Loptr
International Hazard
    
Posts: 1348
Registered: 20-5-2014
Location: USA
Member Is Offline
Mood: Grateful
|
|
Quote: Originally posted by zed  | P2O5?
Well, you probably can't buy it at Ace hardware, but some pyrotechnics suppliers might have it.
It isn't restricted (I don't think); it's just a fussy reagent must be handled carefully.
Available on both Ebay and Amazon, at an outrageous
price.....https://www.ebay.com/itm/Phosphorus-pentoxide-99-5-Analytical-Reagent-ACS/273326700570?hash=item3fa38a781a:g:fJUAAOSwc~NbNQ0H:rk:1:pf:0
But, cheaper elsewhere.
[Edited on 16-10-2018 by zed] |
I never understood why a pyrotechnic supply would have it.
"Question everything generally thought to be obvious." - Dieter Rams
|
|
|
zed
International Hazard
    
Posts: 2285
Registered: 6-9-2008
Location: Great State of Jefferson, City of Portland
Member Is Offline
Mood: Semi-repentant Sith Lord
|
|
Possibly because weird science people like us, might like to buy some (maybe a lot), and the business owners would make money thereby?
"Bobby you're ten years old now, and you are working, so I guess that makes you a man. As long as you have the money, I guess I can sell you a pound
of Potassium Cyanide, if you say you need it. None of my business what you do with it, but be careful. OK?" " Huh? You would like shot of whisky
for the road?" "Sure..... I guess so, but that will be an extra 10 cents!"
|
|
|
clearly_not_atara
International Hazard
    
Posts: 2834
Registered: 3-11-2013
Member Is Offline
Mood: Big
|
|
If I were to guess, it probably increases the sensitivity of some oxidizers, like nitrates and perchlorates, by producing the corresponding anhydrides
N2O5 and Cl2O7, both highly unstable.
|
|
|
itsafineday
Hazard to Self
 
Posts: 89
Registered: 5-1-2019
Location: "...in them thar hills."
Member Is Offline
Mood: Feelin groovy
|
|
Is this good stuff or no?

|
|
|
mayko
International Hazard
    
Posts: 1218
Registered: 17-1-2013
Location: Carrboro, NC
Member Is Offline
Mood: anomalous (Euclid class)
|
|
It's probably good for phosphorus-depleted soil, but it's not actually a bag of phosphorus pentoxide. The MSDS lists the ingredients as mostly calcium
phosphates:
http://www.kochfertilizer.com/pdf/KFB_TSP_GHS_EN_09Nov2012.p...
The analysis label probably reports the content in terms of P2O5 because of the analytical technique used, in the same way that bleach concentration
is sometimes reported in terms of elemental chlorine. If you can figure out that technique, it might help you find a path to P2O5.
al-khemie is not a terrorist organization
"Chemicals, chemicals... I need chemicals!" - George Hayduke
"Wubbalubba dub-dub!" - Rick Sanchez
|
|
|
Tsjerk
International Hazard
    
Posts: 3037
Registered: 20-4-2005
Location: Netherlands
Member Is Offline
Mood: Mood
|
|
0-45-0 just tells you how much P by weight there is in the fertilizer. Nothing more, nothing less.
|
|
|
| Pages:
1
2
3
4
5 |