| Pages:
1
2
3
4
..
10 |
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
And here it is (picture).
Here is the result of the first step. The V2O5 is on the right, the NH4VO3 is on the left. Beautiful.


My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
vulture
Forum Gatekeeper
    
Posts: 3330
Registered: 25-5-2002
Location: France
Member Is Offline
Mood: No Mood
|
|
Axehandle, where did you get the V2O5 and how much did it set you back?
One shouldn't accept or resort to the mutilation of science to appease the mentally impaired.
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
One of the 3 major ceramics suppliers in Sweden, the one based in Stockholm.
0.1kg cost me 207SEK including freight and VAT. That's about 20 Euro, I think.
Took me forever to find someone having it. The guy at the other end of the phone said that the can was very old, and I was the first to order some for
as long as he could remember.
I found 1 (one) supplier in the US, but the price was the same, the freight would have cost more, and they required registration (including my credit
card number!).
I found 0 suppliers on the European continent.
It's an elusive chemical, and I should probably stock up, before it too gets banned.
I don't know if they ship overseas.
They have a web site... let's see, yes, here:
http://user.tninet.se/~svq845w/
[Edited on 2004-3-4 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Found phosphate bonded cement for plugs.
The first dental lab I called today offered me some for free, and if I wanted more, they would sell it to me for what they paid for it. I'm happy
now. I asked what it cost, the guy replied "next to nothing".
Edit: Btw, the excuse I used was "casting mold for aluminum".
Now I only need some rockwool, and some more ammonia as NH4VO3 solvent, then I'm all set, apart from the thermostat thing.
[Edited on 2004-3-4 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
Tacho
National Hazard
   
Posts: 582
Registered: 5-12-2003
Member Is Offline
Mood: No Mood
|
|
Phosphate cement.
I´m glad you foud it. I hope it suits your needs.
I had such a hard time trying to make a good material for high temperatures before I found it! All sorts of messy mixtures with sodium silicate,
metal oxides, carbonates....eechh!
I just could not find information on the chemistry of its hardening. I’m not really interested in the chemistry itself, but on whether it hardens by
hydratation or not.
A plaster without molecules of water could be a nice insulator for jacobs ladders.
I posted this in “general chemistry” but nobody seems to know.
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
| Quote: |
I posted this in “general chemistry” but nobody seems to know.
|
I'll find out. I just got 500g of phosphate bonded cement from a dental lab, for FREE!
   
The broschure says you have to "burn it out" to remove the water. The process won't expand nor constrict the material.
Edit: And if you mix it with water instead of a special fluid he also offered me, it has a heat expansion of 0.58% maximum.
The lab guys say they regularly cast gold using it. So I suppose it's good up to 1800-ish C.   
[Edited on 2004-3-4 by axehandle]
[Edited on 2004-3-4 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
NH4VO3
I wasn't too happy with the colour of the salt, it was slightly yellowish. It could be due to impurities in the V2O5, but it could also be that I
didn't use enough an excess of ammonia.
So now I'm repeating the process, using TWO litres of ammonia. Ouch, my damned sinuses, and alas, my nose, and hope hope hope the neighbours
won't notice the odor.
And I'm going to have to sit in it until it's boiled in. Oh, the fun I have.
Well, I have rockwool now as well. Only thing lacking is Glaubersalz. I suspect it can only be bought as "foot bath salt". I have to find a
way to refine it (i.e. destroy the perfumes and colour agents and other shit).
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
<b> Oh Holy Shit this stinks so bad I want to jump out the window from the 7th floor </b>
I find it ironic right now that my beloved government has banned the sale of even dilute sulfuric acid, while I can cheerfully buy saturated ammonia
(aq) by the tonnes with no questions asked. And acetone. And diethylether. And hydrocloric acid. And ammonium nitrate. And calcium nitrate. And
potassium nitrate. And sodium nitrate. The list goes on....
[Edited on 2004-3-4 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
rikkitikkitavi
Hazard to Others
  
Posts: 192
Registered: 17-6-2002
Member Is Offline
Mood: No Mood
|
|
in princip it would not be so hard to come by battery acid, if you knew someone who owns a gas station...
/rickard
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Well, I unfortunately don't. Besides, I want a limitless supply of 98% H2SO4. And it's more fun to make it.
Edit: Seems most of the NH3 is boiled off now. The air is becoming breathable. Aaaaah.
[Edited on 2004-3-4 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Now. Finally. But the damned thing began to spatter as it thickened, and covered both me and my stove as well as some of my cookware with toxic NH4VO3

I estimate a 1/3 loss, but what remained should be more than enough for at least 3 catalyst tubes.
But I hate getting poison spilled all over me.
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
<b>Fucking</b> stove. Next thing I build will be a proper heating mantle, provided I can find glass fibre tubing.
Damned, NH4VO3 on the skin itches like hell. Don't try it at home, kids... 
And YES, Mr. Marvin, I rinsed and cleaned the exposed skin immediately, and I'm in no fucking way encouraging people to repeat the fucking
accident.
[Edited on 2004-3-4 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Log with pictures.
I have a construction log with some very uninteresting pictures up now, for those interested:
http://species8472.dyndns.org/so3/so3.html
Edit: Oh, this is my 0x100th post! This calls for a celebration!
[Edited on 2004-3-5 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Is this right?
Could THIS really be the colour of the finished catalyst??? I thought it was supposed to be yellow, not reddish-brown????
[Edited on 2004-3-5 by axehandle]
Edit: Apparently it's normal for a catalyst to change color both during preparation and in use. I'll use it.
[Edited on 2004-3-8 by axehandle]

My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
A very interesting concept just struck me. Why heat the catalyst tube? The process of burning the sulfur should give very hot SO2 as a result, which
would then heat up the catalyst automagically. But I'm probably missing something really obvious here....
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
Geomancer
Hazard to Others
  
Posts: 228
Registered: 21-12-2003
Member Is Offline
Mood: No Mood
|
|
The problem, as usual, is control. You don't want the temperature at the catalyst to rise too high, because the yeild decreases with temperature.
I still think that, even on a small scale, the catalyst bed should be able to keep itself more than warm enough if properly designed. Moreover, it is
advantagous to decouple the parts of the process as much as posible, so you don't need to optimize everything at once.You may want to warm your
feed a bit to achieve ignition, though an electric heater has more versatility. If I ever get around to this, I think I'd cool the feed and not
use a heater.
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Yes! Geomancer, that's exactly how also I think! There are 3, possibly 4 variables involved in the control of the SO2 burner exhaust temperature:
1) Air inlet pump speed.
2) Microwave power.
3) Insulation around the catalyst tube.
4) Air inlet air pre-heating (spin a dimmer
controlled heating wire coil around the pipe) or pre-cooling (wrap a wet towel around the pipe).
If all these are kept so that the catalyst temperature is keeps itself at 400-500C _most_ of the time, there should be no need for spinning a complex
heating wire around the catalyst tube.
With a bit of luck (now I'm being optimistic) it should work.
Edit: Btw, my burner is almost done. An old coffee machine glass pot now has a new task in life.... 
Edit2: Fucking hell. Cracked the pot. Ah well, I'll buy a real E-flask prerably with NS-coupling today, then cast a plug with integral air in/out
pipes using phosphate bonded cement.
Just as good the damned thing cracked, in retrospect I doubt it would have coped with (what can it be? 500C?) temperature shock of microwave heated,
burning sulfur.
[Edited on 2004-3-9 by axehandle]
[Edited on 2004-3-9 by axehandle]
[Edited on 2004-3-9 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Might be of interest,
especially for you, Rickard. It seems Melitta brand coffee pots are made out of borosilicate glass.
I just put the remains of my cracked (by a screwdriver impact accident) pot inside the top of my propane furnace and turned it on to full blast.
That's from 26C (my room temperature) to about 900C in a couple of seconds. I let the burner run for about a minute, then I turned it off. The
glass pot remained <b>unscathed</b>.
My conclusion is that a Melitta brand coffee pot can be used as a sulfur burner, given this design (ASCII art):
/
/
| |
| |
-------
Edit: My drawing got fucked by the board code, but you'll all probably get the idea anyway.
As you can see, the converging/diverging top lends itself easily to a gypsum/cement/whatever cast unremoveable plug, with pipes going in and out, and
some sort of re-sealable fuel addition hole.
And they only cost about 50 SEK each, much less than an NS-topped E-flask of borosilicate glass...
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
Tacho
National Hazard
   
Posts: 582
Registered: 5-12-2003
Member Is Offline
Mood: No Mood
|
|
I was planning to post a precise description of a practical thermostat when I had all the details figured out, but there has being a lot of talk about
temperature control here.
I have spent some time trying to figure out a way to make a cheap, easy to make, thermostat to keep the tube furnace at 450º.
I studied three options. I believe the first is the most attractive, although I have never seen it implemented anywhere.
MOLTEN SALT THERMOSTAT.
I am still doing experiments on this. I intent to post a detailed practical project soon.
The idea is that a salt is much more conductive molten than solid. So, a capsule filled with salt has two electrodes, when the temperature of fusion
is achieved, the increase in current triggers off the power. A few observations must be made:
1) Very low current should pass through the salt, to prevent electrolysis that would corrode the electrodes and produce gas. Therefore, an electronic
trigger circuit should be used.
2) The eutectic mixture should melt at a lower temperature, I estimate 440ºC, to compensate for thermal inertia and heat of fusion of the salt.
3) Salt hydrates should be made anhydrous before they are put in the capsule.
4) Since the salts mixtures used to obtain a 440ºC are hygroscopic (if not deliquescent), the capsule should be closed to the atmosphere. On the
other hand, the heating produces increase in pressure due to inevitable (to the amateur) residual water and electrolysis produces a bit of gas, no
matter how low the current. How to solve this? The best I could come with is to connect the system to a rubber bladder like a balloon, or better yet,
a condom. Kind of a joke, but I am still working on it.
5) To find the appropriate eutectic mix:
http://ras.material.tohoku.ac.jp/~molten/molten_eut_query1.p...
I have just begun to test mixes of NaCl, KCl, ZnCl2 and MgCl2.
RTD (RESISTIVE TEMPERATURE DEVICE)
Thermistors could be used here, with the bridge circuit proposed, if you can find a cheap one rated 450ºC. IF YOU FIND IT, LET ME KNOW! The circuit
is ready and it will save me a lot of time.
RTD are just long wires commonly made of platinum, copper or nickel. I add a link to a temperature sensing technologies text that I consider very
good.: http://www.microchip.com/download/appnote/analog/adc/00679a....
There you find a table saying that a platinum wire increases its resistance by 38,5 ohms for every 100ºC of temperature change. An electronic circuit
called a wheatstone bridge connected to an operational amplifier configured to act as a comparator triggers off your power when a certain resistance
is achieved. This sounds complicated, but is a very simple circuit, just half a dozen components on a prototype printed circuit. I´ll post a picture
ASAP.
The big problem here is that platinum is not an option and you need a lot of thin copper wire to get 100 ohms. You should use about that many ohms for
the sake of accuracy and to prevent large currents on your circuit.
We are talking of 16,5 meters of 42 awg wire or 55,25 meters of 37 awg wire (those are the wires I have in stock). 42 is just too thin, I don´t think
it will resist the harsh conditions of a furnace. 37 seem reasonable, but I can´t think of a practical way of putting 55 meters of wire on a sensor
that goes in a furnace. Remember that the varnish will became a very strange thing at 450º, maybe conductive. You also have to protect the wire
against the SO3.
CURIE TEMPERATURE SENSOR
Uh! I love this one, although I never tried it! The idea is that the magnetic properties of a nucleus made of ferromagnetic matter will change its
inductance properties wildly at it´s curie temperature. The beauty is that only the nucleus goes in the furnace, the coil (or coils, if you are
thinking about making a transformer) goes outside, cool and protected.
Shame the only practical materials are nickel (ct=358ºC) and iron (ct=770ºC)
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Temperature control and its whereabouts.
Tacho, I've decided that it's much smarter to heat the burner exhaust gas, instead of heating the catalyst. There are many reasons:
1) The burner gas consists only of dehydrated air and SO2, so it shouldn't be as corrosive to the heating element.
2) The heater's size can now be accomodated to fit the thermostat and heating element, instead of the otrher way around.
3) The catalyst is V2O5 on a pumice carrier. It's thermal capacity is therefore negligible. The catalyst temperature will probably be roughly
the same as the temperature of the gas passing through it.
I've also decided against using a microwave oven as a burner initializer. The main reason is that I need that oven for food, and am too broke now
to buy a new one... 
So, we're left with one choice: A stand-alone sulfur burner, with a heating element for the combustion gas. From now on it gets easier:
I've identified 5 main physical components in the plant:
1: Air feed pump.
2: Air dessicator (CaCl2 filter).
3: Sulfur burner.
4: Exhaust heater and thermostat unit.
5: Catalyst tube (done, will attach a picture)
6: Absorber tank (simple water bubbler).
7) Absorber tank coolant system. (under
design)
We can now concentrate on (4), the most complex of all the parts. The fact that only dry SO2 and air will pass through it gives us plenty of material
choices. It could be as simple as a concrete box (dehydrated) with a free-hanging NiCr coil, some sort of thermostat, and, say, copper tubing for the
inlet and outlet. <b>Dry</b> SO2 shouldn't be corrosive to NiCr wire. At least, I don't think so. And if it is, it's
probably best to design the heater so that the heating element is easily replaceable.
Brainstorming?
/A

My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
Tacho
National Hazard
   
Posts: 582
Registered: 5-12-2003
Member Is Offline
Mood: No Mood
|
|
| Quote: | Originally posted by axehandle
Tacho, I've decided that it's much smarter to heat the burner exhaust gas, instead of heating the catalyst. There are many reasons:
(snip)
/A |
Too late now.
I'll stick half a dozen thermostats down your thread.
Don't try to stop me.
Lets start with an abandoned Idea:This circuit is to be used with RTD (maybe with thermistors) devices. NOT with the molten salt idea because this
will require a much simpler trigger circuit.
Although I believe the most practical idea for a thermostat will be the molten salt, I post this comparator just in case someone is interested. After
all, I´ve already built it anyway.
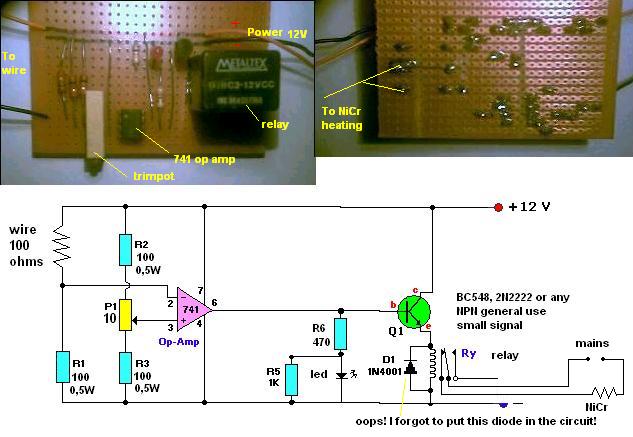
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Even I could build that, on a testing board. Nice circuit.
But I like the molten salt idea. If encased in a glass "bubble" inside the exhaust heater, it should be completely impervious to corrosion.
Edit: Clarification: My new solution does not negate the need for a thermostat, it merely makes the job somewhat easier. (I.e. it doesn't have to
be small).
[Edited on 2004-3-10 by axehandle]
[Edited on 2004-3-10 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
Tacho
National Hazard
   
Posts: 582
Registered: 5-12-2003
Member Is Offline
Mood: No Mood
|
|
THERMOSTATS! THERMOSTATS! THERMOSTATS!
axehandle,
Don´t built the circuit on those solderless breadboards (protoboads)! The comparator depends on fractions of a ohm to work. The resistance of the
contacts of those solderless boards plus oxidation on the wires is enough for erratic behavior. Use solder.
MOLTEN SALT THERMOSTAT
I used a mix of KCl and MgCl2 in 12cm Pyrex test tube. The electrodes are simple copper wires held together by plaster of paris. Results are good:
resistance falls from off-scale to 30 ohms during melting. This test was repeated 4 times.
I added to the tube a bit of zinc I took from a common battery. The melting point of zinc is 420º and it seems to melt along with the salt mix,
although it is not easy to tell when exactly each is molten. This proves that, at least, we are close to 440ºC.
Neither the zinc nor the copper electrode is (are?) attacked by the molten salt.
I repeated the test 24 hours latter and the results were the same (off-scale to 30ohms). This shows that water absorption is not a problem after all.
You may use an open or loosely capped Pyrex test tube as capsule.
I added a trigger circuit that is very simple and I believe your thermostat is ready. I tested it many times and seems to work fine. THIS circuit can
be built on a solderless breadboard, I did. As cheap and simple as possible. Probably needs some heating up to drive away the water after is left
unused for long.
I will be doing some tests during the next days, if something changes I´ll let you know.
Notes:
1) MgCl2 has 6 molecules of water usually bonded to it, so it melts at a lower temperature (dissolves in it´s own water of hydratation), when heated
for the first time. This is good, because it dissolves the KCl and makes a more homogeneous eutectic. Boil away the water and the solid that is left
eventually melts close to 440ºC.
2) Possible mixes(*):
NaCl 56 mol%(37g) + MgCl2 44 mol%(41,89g).................mp= 430ºC
NaCl 56,2 mol%(32,84g) + MgCl2 43,8 mol%(41,70g)......mp= 432ºC
KCl 66 mol% (49,20g) + MgCl2 34 mol% (32,37g)........... mp=430ºC
KCl 66,6 mol% (49,64g) + MgCl2 33,4 mol% (31,8g)....... mp=435ºC
KCl 66,7 mol% (49,72g) + MgCl2 34 mol% (31,7g) ..........mp=437ºC
KCl 65,2 mol% (48,6g) + MgCl2 34,8 mol% (33,13g)....... mp=433ºC
KCl 69 mol% (51,43g) + ZnCl2 31 mol% (42,25g) ............mp=433ºC
(*) I think is clear that the site I linked in the previous post means anhydrous salts, my calculations were based on it, so you have to add the
molecules of water to the weight if using hydatated salt. To save you time: molecular weights: NaCl 58,44; ZnCl2 136,3; KCl 74,54; MgCl2 95,21.
2)My scale is not precise enough to make those different KCl-MgCl2 mixes, so I can only guess my exact melting point.
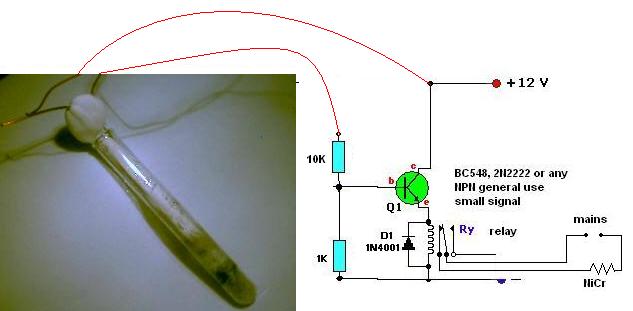
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Beautiful!!!!
And the best part is.......: I have both KCl, metallic zinc and HCl!
I'm concerned about the response time though. Probably best to use a metallic container. Aluminum has the best heat conductivity, and I can
easily cast a probe-like thing.
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
axehandle
Free Radical
    
Posts: 1065
Registered: 30-12-2003
Location: Sweden
Member Is Offline
Mood: horny
|
|
Exhaust heater design, cross-section.
--------------------------------------------------------------------------
Refractory mix
--------------------------------------------------------------------------
Concrete with heating coil + embedded thermostat
--------------------------------------------------------------------------
---> Exhaust flow, pipe length approx. 30cm
--------------------------------------------------------------------------
Concrete with heating coil + embedded thermostat
--------------------------------------------------------------------------
Refractory mix
--------------------------------------------------------------------------
Extremely ugly picture, but you get the idea. The (dehydrated) concrete will act as a heat buffer, so there will be little temperature variance over
time even if the thermostat is slow.
[Edited on 2004-3-11 by axehandle]
My PGP key, Fingerprint 5D96 E09E 365D 1867 2DF5 C2FE 4269 9C19 E079 CD35
\"Verbing nouns weirds the language!\"
|
|
|
| Pages:
1
2
3
4
..
10 |