You can easily adjust lipophilicity of mobile phase by going through the row of methanol-ethanol-propanol-isopropanol-butanol-pentanol...
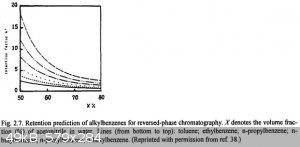
And the retention is usually can be approximately predicted, for example, here is a graph for reverse-pase chromatography of homologs, which show us
the fact that same type of solutes behave similary for the fixed type of stationary and mobile phase, while later can have different composition.
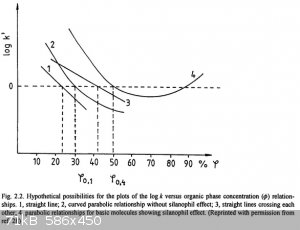
Graph of dependance of logP to composition of solvent is usually either straight line (1 and 3), or downward curve looking like a parabola branch (2),
or falling and then rising curve (4) caused by interaction of silica acidic groups with basic amines. I'd say the reason for the drop in retention
time for mixed solvent is caused by three components interaction (mobile, stationary phases, solute), when the solvent is capable of occupying the
stationary phase surface just as well, as dissolving the solute. In fact, most of the curves for non-basic compounds are of type 2.
Recently I managed to guess the solvent for chromatography in the very first try, the one that caused an almost complete separation of closely related
amine hydrochlorides on TLC (Rf=0.05 first, Rf=0.3 second, Rf=1.0 third) just by knowing their solubility in the used solvent (chloroform: 96%alcohol
= 1:1), while usual solvents from literature would give me some really shitty separation, like 0.37 vs 0.42 for chloroform-acetic acid-water.
The guess-and-try method is even more unrelaible because of the need for mixture of solvents. Chloroform is needed to separate the sample
constituents, but chloroform alone can't efficiently elute the sample, thus leaving everything at start. Pure alcohol would also work for my case, but
give slightly worse separation.
Usage of a sinlge solvent might be advantegous e.g. when you separate basic amine from nonbasic amine: basic amine has higher affinity for stationary
phase, thus higher retention time, but if you try to use a mixture of organic-protic solvent, the retention time of basic amine will be lowered so
much it become the same as retention time of nonbasic amine, because the water negated the sample-silica interaction, making both amines equal.
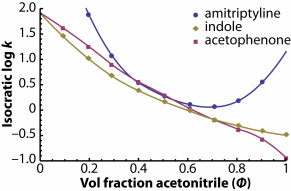
The example on the picture shows the convenience of separation of the three compounds using a pure acetonitrile, which causes the sample compounds to
completely interact with stationary phase active sites. Amitriptyline has extremely low water solubility, thus high retantion time for pure water.
It could be analyticaly deduced even for unknown compounds, the only thing I need to know is that one compounds is completely neutral, the other is
slightly basic, and the third is notably basic.
If I know the compounds have different octanol-water partition coefficients, I could blindly use alcohol (or water for low logP compounds), which
would give a good separation most of the times.
I believe that just by knowing interactions of eluent-stationary phase, eluent-elute, and elute-stationary phases I can approximately pretict the
separation with a good rate of success. But I cannot find much guides in my objective.
|