phendrol
Harmless

Posts: 34
Registered: 13-6-2012
Member Is Offline
Mood: sock puppet
|
|
Quantitative test of a sample of ostarine
Hi!
I have a sample which is suppose to be ostarine of about 99% purity.
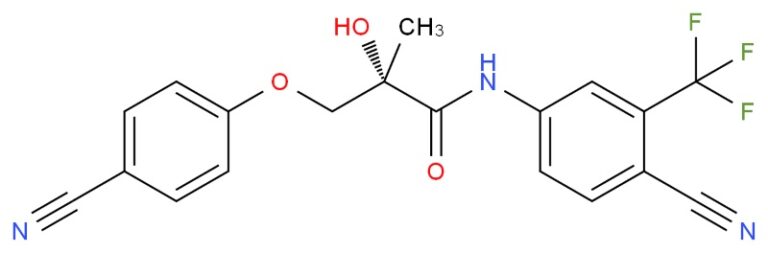
I want to verify it but lab expertise is very expensive. Can you guys give me some advice?
I was thinking to test the solubility and test for the presence of functional groups. Can someone suggest particular tests and reagents for the above
functional groups?
Any advice appreaciated!
|
|
|
Fery
International Hazard
    
Posts: 1010
Registered: 27-8-2019
Location: Czechoslovakia
Member Is Offline
|
|
Hi, not specific test but here I found m.p. 132-136 C
https://en.wikipedia.org/wiki/Enobosarm
|
|
|
Pumukli
National Hazard
   
Posts: 696
Registered: 2-3-2014
Location: EU
Member Is Offline
Mood: No Mood
|
|
I'd try to test for fluorine. Sample digested by alkaline hydroxide and then testing / measuring the freed fluoride ions.
|
|
|
phendrol
Harmless

Posts: 34
Registered: 13-6-2012
Member Is Offline
Mood: sock puppet
|
|
Any reference to the test for fluorine?
|
|
|
AvBaeyer
National Hazard
   
Posts: 647
Registered: 25-2-2014
Location: CA
Member Is Offline
Mood: No Mood
|
|
Fery's suggestion of the melting point is the first step to follow up on. If that checks you are ok if your purity estimate is valid. If the mp is a
bit off, recrystallize and try again. Your main problem with the mp is if the racemate melts differently from a pure enantiomer as you are dealing
with a compound having an asymmetric center. Do you know anything about the chiral purity of the sample? The melting points of the racemate (R/S)and
the resolved isomers (R or S) are likely known providing you with a challenge to find out.
There is no point initially to test for fluorine as that is much more difficult than suggested above. Testing for functional groups is not gong to be
easy. You need to consult a good lab manual on organic qualitative analysis which will provide you with procedures.
BTW, I would not mess with this stuff.
AvB
|
|
|