| Pages:
1
..
14
15
16
17
18
..
25 |
The_Davster
A pnictogen
      
Posts: 2861
Registered: 18-11-2003
Member Is Offline
Mood: .
|
|
For sure, especially if you have seen alibaba prices 
But those 7.5g, turned into a nitrotetrazolate primary would last...if you have a secondary in mind, not so much.
|
|
|
snooby
Hazard to Self
 
Posts: 88
Registered: 24-5-2013
Member Is Offline
Mood: No Mood
|
|
Can someone plz check my calculation I mentioned 3 posts above, since I dont want to waste a lot of holy grale chems!!
|
|
|
Dornier 335A
Hazard to Others
  
Posts: 231
Registered: 10-5-2013
Location: Northern Europe
Member Is Offline
Mood: No Mood
|
|
Engager stated 0.00689 mol NaNTZ. The molar weight of NaCN5O2*2H2O is 173.06 g/mol. 0.00689 mol*173.06 g/mol = 1.192
g.
The molar ratio of the copper complex to sodium nitrotetrazolate is 2:1, which I don't really see the point of. But I think you should follow
Engager's instructions.
|
|
|
snooby
Hazard to Self
 
Posts: 88
Registered: 24-5-2013
Member Is Offline
Mood: No Mood
|
|
Well I calculated the same tnx: 3.5 grams of carbonato triammine copper(II) nitrate for every 1.91 grams of NANTZ.
With BNCP it is the opposite: 1.4 grams of cobalt stuff for every 2.6 grams of NANTZ.
Since NANTZ is harder to make, maybe the copperstuff is better variant to make! You need a lot less of NANTZ!
|
|
|
snooby
Hazard to Self
 
Posts: 88
Registered: 24-5-2013
Member Is Offline
Mood: No Mood
|
|
I made BNCP and NaCUNTZ. I was able to set off BNCP with a relatively simple primer such as mealpowder. However, NaCUNTZ seems to need a hotter
primer. I think I can get 5/10 in my blastingcaps with NaCUNTZ. So, I am looking for a decent priming composition. I have tried 80 leadtetroxide/20
silicon and this one burns veryy fast (flashpowder like). Mayb you guys have some suggestions for a very hot primining composition.
|
|
|
Dornier 335A
Hazard to Others
  
Posts: 231
Registered: 10-5-2013
Location: Northern Europe
Member Is Offline
Mood: No Mood
|
|
I synthesised a small amount of C2H4N14 bis-diazotetrazolylhydrazine, or as I prefer to call it,
ditetrazolylhexaazadiene.
When I dripped almost colourless diazotetrazole solution into hydrazinium chloride solution, it turned pink and then a fluffy yellow precipitate
formed. It hydrolyzed again before I could vacuum filter it. After a few days tiny dense grains of a yellow compound had precipitated. A single of
those grains, approx 50 micrograms detonates violently enough to rip a hole though aluminium foil when heated slowly. Still, it's not the unequivocal
primary I though it would be. A few milligrams only puff and spread out the pile when ignited. Strongly pressed it is extremely brisant.
Density was calculated to approx 1.75 g/cc by group addition method, similar to 5-azidotetrazole (1.72 g/cc), 1-diazidocarbamoyl-5-azidotetrazole
(1.72 g/cc) and 1,1'-azobis(tetrazole) (1.77 g/cc). Enthalpy of formation is most likely between +1300 and +1500 kJ/mol based on its structure and
similar compounds. This gives it a detonation velocity of around 9300 m/s.
Here's my video about it: https://www.youtube.com/watch?v=qVZT5aR3ih0
|
|
|
Rosco Bodine
Banned
Posts: 6370
Registered: 29-9-2004
Member Is Offline
Mood: analytical
|
|
DBX-1
One of the inventors passed this along to me. Sharing is caring. 
I get a real chuckle knowing that some of the leading high technology contributors of this world are associated with this ..er ..uh .."amateur"
science forum. 
Attachment: US20150239910A1 METHOD FOR PREPARATION OF A LEAD-FREE PRIMARY EXPLOSIVE.pdf (46kB)
This file has been downloaded 1045 times
|
|
|
Jimbo Jones
Hazard to Others
  
Posts: 102
Registered: 15-10-2009
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by Rosco Bodine  | One of the inventors passed this along to me. Sharing is caring. 
I get a real chuckle knowing that some of the leading high technology contributors of this world are associated with this ..er ..uh .."amateur"
science forum. 
|
You can bet on it...
|
|
|
Rosco Bodine
Banned
Posts: 6370
Registered: 29-9-2004
Member Is Offline
Mood: analytical
|
|
guanazole reaction revisited
From about 7 years ago to again about 4 years ago and later a couple of years ago the subject of guanazole has received some attention. This post by
Engager links back to the original discussion in another thread which is continued here in this thread.
http://www.sciencemadness.org/talk/viewthread.php?tid=8144&a...
I have some additional information snippets which are tending to support my earlier idea of synthesizing guanazole using the patent process of
US2648671 which is reaction of molar equivalents of cyanoguanidine with hydrazine dihydrochloride in aqueous suspension to provide a quantitative
yield of guanazole.
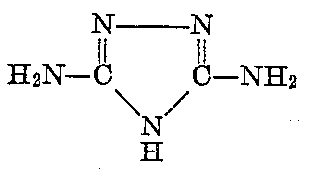
I still speculate it may be possible to proceed without isolation of the guanazole to diazotize and produce the dinitro derivative,
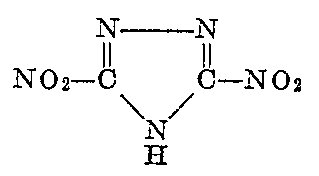
or to produce the 50/50 mixture of the dinitro derivative with 5-nitrotetrazole via Sandmeyer reaction condition.
I think this may possibly be the easiest route to a tetrazole or more precisely to a mixture or possible double salt that is 50% 5-nitrotetrazole with
the other 50% being 3,5-Dinitro-1,2,4-triazole according to the method of US3054800.
The best strategy for accomplishing this synthetic route is not yet identified and there would seem to be variations on the approach possible, which
could involve isolation of a guanazole derivative as an intermediate, either a guanazole double salt complex with copper sulfate, or if it exists even
better would be a double salt complex of guanazole with copper nitrate.
An additional possibility is the formation of a guanazole nitroso derivative which may be a not previously identified intermediate for either
3,5-Dinitro-1,2,4-triazole, or possibly an intermediate in the patent process leading to the 50/50 mixed product. I believe that a possible isolation
of the copper salt or ammonium complex copper salt, or double salt could be done. Or it may be possible to separate the 50/50 mixture into its
components, depending upon how the process is done.
Does anyone have any information as to whether or not guanazole forms a double salt with copper nitrate as does form with copper sulfate? Further,
does anyone have information about the nitroso derivative of guanazole, which would possibly be guanazole oxime? See the article attached page 518.
Also attached is an abstract for a Russian article which is not available online, but which may be helpful.
I am still studying and reviewing what are available references so I am sharing what I accumulate on this potentially useful synthetic route and its
possible products. The silver and the copper salts would seem to be of interest. And of course there are many possible double salts not yet
identified.
Attachment: from JCS page 517 guanazole.pdf (215kB)
This file has been downloaded 855 times
Attachment: rjapchem0839_abstract Synthesis 3,5-dinitro-1,2,4-triazole, Russian Journal of Applied Chemistry, Vol. 73, No. 5, (2000) (3kB)
This file has been downloaded 987 times
Attachment: guanazole pages from PATR Vol. 5 D(cont.)-E.pdf (148kB)
This file has been downloaded 1149 times
Attachment: guanazole page PATR Vol. 6 E(cont.)-G.pdf (92kB)
This file has been downloaded 768 times
Attachment: US2648671 PREPARATION OF GUANAZOLE.pdf (84kB)
This file has been downloaded 1035 times
Attachment: US3054800 50-50 mixture of dinitrotriazole and 5-nitrotetrazole via Sandmeyer reaction on guanazole.pdf (130kB)
This file has been downloaded 731 times
Attachment: preparation and characterization of 3,5-dinitro-iH-1,2,4-triazole.pdf (1.5MB)
This file has been downloaded 1212 times
Attachment: prep-chem.pdf (69kB)
This file has been downloaded 716 times
Attachment: Synthesis, Properties and Performance of the High Explosive ANTA.pdf (555kB)
This file has been downloaded 1056 times
Attachment: Pages from dubious Ledgard.pdf (203kB)
This file has been downloaded 815 times
There has been other interest in guanazole conversion to
3,5-Dinitro-1,2,4-triazole
https://www.youtube.com/watch?v=IxEpkA1V46k
<iframe sandbox width="640" height="360" src="https://www.youtube.com/embed/IxEpkA1V46k?rel=0" frameborder="0" allowfullscreen></iframe>
[Edited on 9/25/2015 by Rosco Bodine]
|
|
|
Mr.Greeenix
Harmless

Posts: 40
Registered: 8-4-2015
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by snooby  | | The DBX-1 turned almost black in the end. When dry it became little chunks of DBX-1 who where impossible to be powdered. They were quite sensitive for
impact but did not work when ignited by fuse unfortunately. The synth I used was whithout the use of NaNTZ. Maybe this synth was not so good after
all. I now have an amount of cupric(II)cloride and I will try the synth again in a while. I indeed decided to put away the stuff. Have you tried DBX-1
yourself btw? |
I tried recently it. With the method Rosco Bodine posted here http://www.sciencemadness.org/talk/viewthread.php?tid=8144&a...
The synthesis worked just fine even when I haven't reach the temp. of 100°C at the end (had about 80°C)
My DBX-1 was first reddish but finally after it was fully dry it also gets its almost black apperance. These chunks were
difficult to powder but possible. I broke them up carefully. Finally I got the rusty colour stated in the literature. Also much more flame sensitiv
and gives a greenish flame when ignited.
____
Only fictive!
[Edited on 26-9-2015 by Mr.Greeenix]
|
|
|
Mr.Greeenix
Harmless

Posts: 40
Registered: 8-4-2015
Member Is Offline
Mood: No Mood
|
|
I synthetised 5-Aminotetrazole two times now.
The first time small scale with the method posted by Engager. The yield was ok and the product was very pure.
But now I scaled it up a little bit.
Approximately 10g Aminoguanidin bicarbonat ; 200ml 15% sulfuric acid ; 10g Nano2
and much sodium carbonate for the PH.
The problem is that I had a massiv yield of about 100g. I suppose that this is too good to be true
After cooling there were only crystals left in the beaker no more fluid, so I guess that all impurities also crystallized.
The Ph of my "5-Aminotetrazole" is neutral.
Is someone able to explain this yield?
Does someone know how to remove the sodium carbonat or sodium sulfat (Cause I neutralized with sulfuric acid) in my 5-Aminotetrazole. Suitable
Recrystallization?
(Video to this method: https://www.youtube.com/watch?v=oTjgHo-EdXY) he also had a massiv yield but I halved the amount of chemicals used.
Thank you 
|
|
|
Boffis
International Hazard
    
Posts: 1867
Registered: 1-5-2011
Member Is Offline
Mood: No Mood
|
|
@Mr.Greenix it sounds like you have much sodium sulphate hydrate in there! 5-aminotetrazole crystallises as the monohydrate so you should get about
7.5g at 100% yield but you usually only get about 75% with the Thiele method.
Try treating the product with dilute HCl to ensure that it is the free tetrazole derivative, then leach with water until you have got rid of most of
the sodium salts and then recrystallise from water. 5-aminotetrazole is very soluble in hot water but sparingly soluble in cold water and salt
solutions so the recovery during recrystallisation is usually good.
By the way, always allow the filtrate to stand for several days or a week in contact with a few grains of the initial product and also stir
occasionally making sure you rub the vessel walls occasionally. 5-aminotetrazole has a terrible tendency to supersaturate.
PS just because somebody makes a youtube video it doesn't mean they know what they are doing!
|
|
|
Rosco Bodine
Banned
Posts: 6370
Registered: 29-9-2004
Member Is Offline
Mood: analytical
|
|
Here is a doctoral dissertation of interest
694 pages - 35.1 MB
Advanced Energetic Materials Based on 5-Aminotetrazole - Synthesis, Characterization, Testing and Scale-up - Stierstorfer_Joerg.pdf
https://edoc.ub.uni-muenchen.de/12994/1/Stierstorfer_Joerg.p...
http://www.hedm.cup.uni-muenchen.de/personen/staff/stierstor...
http://www.hedm.cup.uni-muenchen.de/index.html
Also another interesting and more recent doctoral dissertation for which a link was posted earlier, and then disabled temporarily due to a
confidentiality about a pending patent application, is again available for download since the patent has issued for the DBX-1 process described
therein.
http://www.sciencemadness.org/talk/viewthread.php?tid=8144&a...
The dissertation is
Advanced Energetic Materials - Piercey_Davin.pdf
linked here again
https://edoc.ub.uni-muenchen.de/15389/1/Piercey_Davin.pdf
[Edited on 3/26/2016 by Rosco Bodine]
|
|
|
octonitrocubane
Banned troll

Posts: 9
Registered: 17-3-2016
Member Is Offline
Mood: No Mood
|
|
Anyone for 1,6-ditetrazolylhexaaza-1,5-diene? Dornier335A has it on Youtube. VoD 9300 ms. Sensitivity is, of course, just about unparalleled. Formula
C2H4N14 - it contains 87.5% nitrogen by weight.
|
|
|
Rosco Bodine
Banned
Posts: 6370
Registered: 29-9-2004
Member Is Offline
Mood: analytical
|
|
Quote: Originally posted by octonitrocubane  | | Anyone for 1,6-ditetrazolylhexaaza-1,5-diene? Dornier335A has it on Youtube. VoD 9300 ms. Sensitivity is, of course, just about unparalleled. Formula
C2H4N14 - it contains 87.5% nitrogen by weight. |
Here's the video
https://www.youtube.com/watch?v=WowrwV-0ca8
<object width=640 height=360><param name="movie"
value="http://www.youtube.com/v/WowrwV-0ca8?version=3&autoplay=0&showinfo=1&modestbranding=1&controls=1&theme=dark&vq=hd720&am
p;hl=en_US&rel=0"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess"
value="always"></param><embed
src="http://www.youtube.com/v/WowrwV-0ca8?version=3&autoplay=0&showinfo=1&modestbranding=1&controls=1&theme=dark&vq=hd720&
hl=en_US&rel=0" type="application/x-shockwave-flash" width=640 height=360 allowscriptaccess="always"
allowfullscreen="true"></embed></object>
https://www.youtube.com/watch?v=qVZT5aR3ih0
<object width=640 height=360><param name="movie"
value="http://www.youtube.com/v/qVZT5aR3ih0?version=3&autoplay=0&showinfo=1&modestbranding=1&controls=1&theme=dark&vq=hd720&am
p;hl=en_US&rel=0"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess"
value="always"></param><embed
src="http://www.youtube.com/v/qVZT5aR3ih0?version=3&autoplay=0&showinfo=1&modestbranding=1&controls=1&theme=dark&vq=hd720&
hl=en_US&rel=0" type="application/x-shockwave-flash" width=640 height=360 allowscriptaccess="always"
allowfullscreen="true"></embed></object>
[Edited on 3/27/2016 by Rosco Bodine]
|
|
|
Dornier 335A
Hazard to Others
  
Posts: 231
Registered: 10-5-2013
Location: Northern Europe
Member Is Offline
Mood: No Mood
|
|
The first link in Rosco's post shows its properties very well, but I'll provide some more information here.
Ditetetrazolylhexaazadiene is described in Engager's Energetic Derivatives of Tetrazole with the name bisdiazotetrazolylhydrazine and
briefly in Fedoroff. It's synthesised by mixing diazotetrazole solution with a solution of hydrazinium chloride and sodium acetate. My product
immediately dissolved/decomposed after forming though, probably due to too basic conditions. Replacing the hydrazinium chloride with hydrazine sulfate
worked well, and sodium acetate buffer doesn't seem necessary, although the product is whiter without.
The product seems stable after drying but rapidly dissolves in basic solutions to give a strongly yellow solution. It hydrolyses slowly in water to
give gas bubbles (nitrogen?) and 5-azidotetrazole. My product is stable in acidic solutions though.
Now comes the fun part: it's more explosive than anyone has probably ever dreamt of. My calculated performance at the measured density 1.8
g/cm3 is 9700 m/s and 420 kbar.
In addition, it's critical diameter is microscopic. I played around with tiny grains under a microscope, trying to make as small flakes as possible. I
got an 18 micron thick flake, approximately 0.4 micrograms, to detonate (it put a dent in the aluminium foil).
This performance is not without downsides of course; it is exceptionally sensitive. Filter papers covered with ditetetrazolylhexaazadiene explode if
ripped (something I discovered without protection...) Light tapping with a hammer sets it off, as does pressing. More videos will come in the future!
[Edited on 27-3-2016 by Dornier 335A]
|
|
|
Rosco Bodine
Banned
Posts: 6370
Registered: 29-9-2004
Member Is Offline
Mood: analytical
|
|
Regarding the aminoguandine bicarbonate precursor for aminotetrazole
A point of interest is a second independent confirmation regarding the pyrolitic decomposition of calcium cyanurate to pure calcium cyanamide. I
think Davin first reported this does in fact work even as a "campfire" cyanamide produced in the hot coals of a wood fire kind of adapted process so
long as there is venting of the byproduct gases from the heated to orange heat container allowed, but not allowing air ingress into the reaction
mixture.
It looks like maybe a hacksaw cut was made to defeat the sealing of the threads and allow controlled venting for the "bomb" used for the pyrolysis
which is good technique. A small press fit "freeze plug" type of safety overpressure blowout plug is a good idea also in the top end cap for a
reactor like this in case venting is ever obstructed to prevent pressure buildup in the reactor. Probably a good easy safety vent providing a "gravity
seal" would be to use a 3/8" clevis pin dropped into a 3/8" hole drilled into the end cap, with the loose seal at the shoulder being sufficient. The
shaft of the clevis pin could be scored with a diagonal groove cut using a hacksaw, and if pressure from venting raised the clevis pin to the cut
groove it should regulate there by gravity like a pressure cooker regulator, and function as a loosely fitted check valve and regulator.
https://www.youtube.com/watch?v=AF11Xuh0SaQ
<object width=640 height=480><param name="movie"
value="http://www.youtube.com/v/AF11Xuh0SaQ?version=3&autoplay=0&showinfo=1&modestbranding=1&controls=1&theme=dark&vq=hd720&am
p;hl=en_US&rel=0"></param><param name="allowFullScreen" value="true"></param><param name="allowscriptaccess"
value="always"></param><embed
src="http://www.youtube.com/v/AF11Xuh0SaQ?version=3&autoplay=0&showinfo=1&modestbranding=1&controls=1&theme=dark&vq=hd720&
hl=en_US&rel=0" type="application/x-shockwave-flash" width=640 height=480 allowscriptaccess="always"
allowfullscreen="true"></embed></object>
[Edited on 3/27/2016 by Rosco Bodine]
|
|
|
octonitrocubane
Banned troll

Posts: 9
Registered: 17-3-2016
Member Is Offline
Mood: No Mood
|
|
Having just synthesised five grams, its safe to say that I will never touch it again. One flake on the floor nearky gave me a heart attack. 4 grams
contained with aluminium foil within a plastic bag lit at the bottom of the water bottle absolutely shred it. Despite being able to isolate ~10 micron
flakes, it has yet to not detonate...
|
|
|
Rosco Bodine
Banned
Posts: 6370
Registered: 29-9-2004
Member Is Offline
Mood: analytical
|
|
early thread missing images archived
Some of the early pages of this thread have dead linked missing images so attached is an old backup pdf which has viewable most of the missing files
Attachment: Tetrazole thread archival images.pdf (2.4MB)
This file has been downloaded 1496 times
|
|
|
Erwinol
Harmless

Posts: 1
Registered: 10-4-2016
Member Is Offline
Mood: No Mood
|
|
http://www.wydawnictwa.ipo.waw.pl/cejem/2-2010/full/Sabate.p...
|
|
|
NeonPulse
Hazard to Others
  
Posts: 417
Registered: 29-6-2013
Location: The other end of the internet.
Member Is Offline
Mood: Isolated from Reality! For Real this time....
|
|
Ok so it has been quite a journey from pool chemicals, urea, sodium bicarbonate and hydroxide , ammonia, hydrochloric, sulfuric, nitric acids copper
sulfate sodium nitrite and anything else I had forgotten but finally I got to make some of the copper nitrotetroazolate primary I had aimed for.
All components were OTC or made from OTC components except the 70% nitric acid( which could easily be synthesised OTC anyway) including the ammonia
which I made from NaOH and ammonium sulfate, the hydrazine, the cyanamide ( my method was shown in video above) which all together formed the basis of
the 5-ATZ and finally the CuNTZ.
Basically I had followed Engagers work here. It's his legacy here it would seem...
I must say though I'm pretty impressed by this compound although it is not the best initiator made from the 5-ATZ and really want to attempt the
silver variant and the Mercury variant especially. This particular salt gave me no troubles synthesising it and I was expecting the micro explosions I
had read about but luckily for me none happened. The only problems were the NOx fuming and thevery slow filtering of the ultra fine green mud.
It seems to have a tiny critical diameter and minute grains go off with a crack when heated on foil putting pin holes in it but with confinement in
foil and heated it gives a pretty loud report and sends silver foil confetti flying. I want to test it a little more but so far I can see why
primaries based on 5-ATZ and its salts are of interest as green energetic materials.

|
|
|
Rosco Bodine
Banned
Posts: 6370
Registered: 29-9-2004
Member Is Offline
Mood: analytical
|
|
Voila! ..... Verdigris Patina Terribilis 
seafoam green with attitude 
The color reminds me of the patina on roofs and statuary

[Edited on 4/24/2016 by Rosco Bodine]
|
|
|
Laboratory of Liptakov
International Hazard
    
Posts: 1386
Registered: 2-9-2014
Location: Technion Haifa
Member Is Offline
Mood: old jew
|
|
roof
Oh my God. On roofs I see Neon - Cu NTZ compounds! Is not necessary huge a complication of synthesis. Much easily is scratching from the roof. Without
acid and others chemical tools. I seems, that only what you needed is knife and ladder. Thanks for info... ...LL ...LL
|
|
|
Dornier 335A
Hazard to Others
  
Posts: 231
Registered: 10-5-2013
Location: Northern Europe
Member Is Offline
Mood: No Mood
|
|
NeonPulse, is that Cu(NTZ)2 or is it the acid copper nitrotetrazole salt? Because that one is supposed to be very
sensitive.
Liptakov, I climbed my local church's roof and found this:

It turned out to be copper(II) 5-azidotetrazolate though! Be careful!
|
|
|
NeonPulse
Hazard to Others
  
Posts: 417
Registered: 29-6-2013
Location: The other end of the internet.
Member Is Offline
Mood: Isolated from Reality! For Real this time....
|
|
It's the acid copper salt. It's colour is much paler than the picture portrayed but the camera seem to get it right. It would be much better if it
had a higher density like crystals rather than the fine fine powder. It doesn't seem overly sensitive but a lot of care is still taken while handling
the dry product. Once it had been dried for weighing I have sprayed a mist of water to dampen it and put it in a container till it's needed.
Is it right that being stored underwater will slowly decompose it? I think I read that in some PowerPoint presentation about green initiators some
time ago.
Wet storage is much preferred to missing fingers and I can easily see a small amount doing a lot of damage to a careless handler.
I know it seemed overly complicated but it's not really.Basically I wanted to see if this could be done using all OTC chemicals and it seems that with
a bit of work and effort it can. However for all that work put in over a few weekends the end result was only a few grams of the green stuff. But
that's enough since a little bit goes s long way.Most people would probably say it's hardly worth the time but hey, I think it's time well spent.
I'll Give the photo another go:

|
|
|
| Pages:
1
..
14
15
16
17
18
..
25 |