| Pages:
1
2 |
homealchemy1
Harmless

Posts: 1
Registered: 5-4-2015
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by Deathunter88  |
The inner glass cracks and water from the jacket flows in to the inner tube. Due to the temperature change it instantly flash boils into steam. The
steam pushes the remaining water back into the boiling flask and the cold water enters the 300+ degrees boiling sulphuric acid. The entire apparatus
blows apart in one of those "Don't pour water into a grease fire" demos except this time it's a huge plume of sulphuric acid vapour and mist. Am I
missing anything? |
Are you basing this on your personal experience or theorizing it? Nurdrage is a veteran not an amateur. I hate when someone pontificate from the
ivory tower. Deathunder88 -- have you tried this distillation using your own method?
The fact is that you must first run the water and as the temp. of the vapor is ramp up gradually, it will condense. that's why its absolutely
important that you run the condenser first with water and slowly increase the temperature of the mantle.
I've tried it on a small scale and its fine. You must not rush this distillation!!! Don't use the three way adapter, use the 75 degree bent adapter
just like Nurdrage did. It took me 5 hrs to do everything and clean up and I got 100ml of clean acid.
[Edited on 5-4-2015 by homealchemy1]
|
|
|
Deathunter88
National Hazard
   
Posts: 519
Registered: 20-2-2015
Location: Beijing, China
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by homealchemy1  | Quote: Originally posted by Deathunter88  |
The inner glass cracks and water from the jacket flows in to the inner tube. Due to the temperature change it instantly flash boils into steam. The
steam pushes the remaining water back into the boiling flask and the cold water enters the 300+ degrees boiling sulphuric acid. The entire apparatus
blows apart in one of those "Don't pour water into a grease fire" demos except this time it's a huge plume of sulphuric acid vapour and mist. Am I
missing anything? |
Are you basing this on your personal experience or theorizing it? Nurdrage is a veteran not an amateur. I hate when someone pontificate from the
ivory tower. Deathunder88 -- have you tried this distillation using your own method?
The fact is that you must first run the water and as the temp. of the vapor is ramp up gradually, it will condense. that's why its absolutely
important that you run the condenser first with water and slowly increase the temperature of the mantle.
I've tried it on a small scale and its fine. You must not rush this distillation!!! Don't use the three way adapter, use the 75 degree bent adapter
just like Nurdrage did. It took me 5 hrs to do everything and clean up and I got 100ml of clean acid.
[Edited on 5-4-2015 by homealchemy1] |
It is speculation, but I don't want to find out if it will actually happen. Besides, its good practice to use an air condenser for any liquid that
boils above 150 degrees C.
|
|
|
Trizocy
Harmless

Posts: 34
Registered: 16-2-2015
Member Is Offline
Mood: No Mood
|
|
| Quote: | Are you basing this on your personal experience or theorizing it? Nurdrage is a veteran not an amateur. I hate when someone pontificate from the
ivory tower. Deathunder88 -- have you tried this distillation using your own method?
The fact is that you must first run the water and as the temp. of the vapor is ramp up gradually, it will condense. that's why its absolutely
important that you run the condenser first with water and slowly increase the temperature of the mantle.
I've tried it on a small scale and its fine. You must not rush this distillation!!! Don't use the three way adapter, use the 75 degree bent adapter
just like Nurdrage did. It took me 5 hrs to do everything and clean up and I got 100ml of clean acid. |
Thanks, but is the hub good enough or do i need to order a heating mantle?
|
|
|
Magpie
lab constructor
    
Posts: 5939
Registered: 1-11-2003
Location: USA
Member Is Offline
Mood: Chemistry: the subtle science.
|
|
I can buy Rooto 96-98% H2SO4 so don't have to suffer the indignity of distilling this dangerous reagent.
Instead of plugging the stillhead with a thermometer adapter use a glass plug. If you can get a tapered glass thermometer insert that would be ideal.
How necessary is temperature indication to this process?
I would definitely avoid using water as coolant - using forced air or nothing sounds like the way to go.
A good quality borosilicate West or Liebig condenser should handle temperature differences well. I have distilled 200°C bp liquids many times with
ice water (don't ask me why the ice - habit, I guess) in my West condenser with no problems.
[Edited on 5-4-2015 by Magpie]
The single most important condition for a successful synthesis is good mixing - Nicodem
|
|
|
Fulmen
International Hazard
    
Posts: 1716
Registered: 24-9-2005
Member Is Offline
Mood: Bored
|
|
I think it's pretty much common sense not using water at these temperatures, especially with stuff like strong acids.
As for the setup wouldn't a column be required for concentrating without too much loss? If so I'd consider using the cooler as an insulated column and
using something like a vigeraux for cooling. The reflux could then be adjusted with airflow through the cooling jacket. Or am I missing something?
We're not banging rocks together here. We know how to put a man back together.
|
|
|
Trizocy
Harmless

Posts: 34
Registered: 16-2-2015
Member Is Offline
Mood: No Mood
|
|
| Quote: | I can buy Rooto 96-98% H2SO4 so don't have to suffer the indignity of distilling this dangerous reagent.
|
Nice for you, but for me i cant import it to my country, really strict when it comes to concentrate sulpuric acid, its on: "watch over list"
Only thing i can get is drain cleaner with 50%-90%, so the only way i can get it concenrated is distiling it.
May someone share a video on youtube, where they use air as coolant at the condenser and not water? Only finding water coolant :/
|
|
|
Trizocy
Harmless

Posts: 34
Registered: 16-2-2015
Member Is Offline
Mood: No Mood
|
|
Was wondering how concentrate can i get the sulpuric acid if i just use a beaker and hub?
|
|
|
DSC_JN
Harmless

Posts: 5
Registered: 3-4-2015
Member Is Offline
Mood: No Mood
|
|
Sulfuric Acid
You might be able to obtain 96% Sulfuric acid by contacting agricultural suppliers, 96% is often used to balance the pH of animal manure. Often it is
possible to obtain a permission/dispensation from the rules depending on usage.
Does anyone here have any suggestions on how to recover and seperate sulfuric acid and nitric acid waste from nitric acid distillation to around 99+%?
Keep in mind that the waste might contain some remains of nitric acid and will have a sulfuric acid concentration of around 70%.
Jeppe Nielsen, Danish Space Challenge (DSC)
EDIT: Our current procedure for waste handling is just to use the slightly diluted acid to neutralize animal manure; a process were any remains of the
nitric acid will improve the manure as a fertilizer.
[Edited on 5-4-2015 by DSC_JN]
|
|
|
radiance88
Hazard to Self
 
Posts: 64
Registered: 18-12-2014
Location: underground volcano fortress
Member Is Offline
Mood: a little less evil than usual
|
|
Guys OP here is 13 years old. I highly doubt having him run through the bureaucratic mill is going to get him the acid that he needs. He definitely
needs to distill/clean it himself.
Btw kudos OP. You've got great English and it's always nice to have members from such distant and unexpected places.
|
|
|
DSC_JN
Harmless

Posts: 5
Registered: 3-4-2015
Member Is Offline
Mood: No Mood
|
|
Unfortunately it's nearly impossible to legally obtain small quantities of certain chemicals (kg or liters), but when you try ordering in bulk (tons
or cubic meters) then it becomes easy to obtain chemicals which are otherwise more or less impossible to get.
Personal experience:
Getting 100-200kg of 99+% Nitric Acid is impossible, but ordering 28 tons is easy. We ended up having to distill 53% nitric acid to 99+% by mixing it
with sulfuric acid (96%) in order to bind the water. That's why I suggested agricultural suppliers, as we had luck obtaining 53% nitric acid and 96%
sulfuric acid from them in bulk scales.
Point of the story, even with legalities in order it's near impossible to obtain small amounts of restricted chemicals, at least at reasonable prices.
If you can, order bulk amounts.
Jeppe Nielsen, DSC
|
|
|
byko3y
National Hazard
   
Posts: 721
Registered: 16-3-2015
Member Is Offline
Mood: dooM
|
|
Wonna tell you something from my experience of H2SO4 distillation.
One of the problems is already discussed here - you can't cool the condenser with water. The other problem I had - leaking acid through the horizontal
socket between stillhead and condenser above 290 C.
At the picture you can see a good example of setup you can NOT use for distilling sulfuric acid above 85%.

Probably if you turn your stillhead 45 degrees so all your sockets will be at least partially vertical - you might be able to avoid horrible acid
smoke. It works better for 60-75 degrees angle on your three-way adapter. (remember, that you cannot use water in the first condenser)
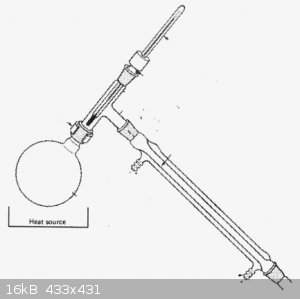
You need a setup with all sockets vertical on the hot side and preferable air cooled, like this one.

and a water-cooled vertical condencer to cool the hot liquid acid coming from the adapter, like in this setup

This way I made 90% concentration and at least 99% pure sulfuric acid that was as clear as distilled water. Would be glad to show you photos of my
setup, but they are bad and won't show you much.
I've never encountered any large amount of water leaking into the hot concentrated sulfuric acid, but I've seen one drop of water falling into the
boiling acid - it made a little explosion. I would not dare to allow any appreciable amount of water to be anywhere near the hot acid.
Once I had a cold acid sucked back into the hot part. It went pretty safe and the distillation ended immediately, but - I've got a stillhead and
condenser destroyed.
The same reason why you should absorb SO3 with sulfuric acid and not with water - because water explosively turns into steam on contact.
|
|
|
Trizocy
Harmless

Posts: 34
Registered: 16-2-2015
Member Is Offline
Mood: No Mood
|
|
Hmm, thanks bykno3y.
I have got a setup like nurdrage now, so gonna try to distill some sulp acid soon.
Got a water pump laying around here, but are skeptical to use it, since most here dont recommend it. Still strange that nurdrage use water with the
condenser like it was no problem.
|
|
|
byko3y
National Hazard
   
Posts: 721
Registered: 16-3-2015
Member Is Offline
Mood: dooM
|
|
I doubt that nurdrage got 98% H2SO4. First, because it lays in the last 20-30% of sulfuric acid. And second, because he uses open receiving flask, and
90%+ sulfuric acid readily absorbs moisture from air.
Also, I forgot to mention that you need a porcelain boiling stones, preferably ones that were already treated with conc sulfuric acid (otherwise some
crap the stones them will come into our acid). I made my from a broken porcelain cup. Porcelain brick also seems to work well.
|
|
|
Mesa
Hazard to Others
  
Posts: 264
Registered: 2-7-2013
Member Is Offline
Mood: No Mood
|
|
Lots of the posts here are drastically underestimating borosilicate(and quartz) glass resistance to thermal stress/shock.
Quote: Originally posted by Marvin  |
You've indicated you can get Quartz glassware in another thread. I suggest a Quartz flask coupled to a Quartz air condenser. The acid will pick up
water from the air. Some form of receiver open to the air but protected by a moisture guard would help. (Do NOT try to seal the system completely).
Edit, I should also add that with an air condensor the acid coming over will be very hot and the receiver should at least be borosilicate, if it it's
jointed glassware then it should probably quartz too to avoid problems.
[Edited on 31-3-2015 by Marvin] |
In what possible scenario would a distillation flask under constant heat from either a hotplate + sandbath or heating mantle would the temperature
differential of the glass get anywhere near the limits required for borosilicate glass to fracture?(which is roughly 160*c)
Extra points if it's explained how you intend to join the quartz flask to the borosilicate stillhead and expect it to have a higher chance of
surviving.
Conditions within a laboratory setup/reaction need to be ridiculously volatile to require the use of quartz glass.
Short of unscrewing the thermometer adapter and pouring water or some organic compound directly into the boiling flask mid-distillation, the only
source of heat in the entire procedure will be the hotplate itself. Unless the laws of thermodynamics changed recently, vaporization doesn't release
heat.
[Edited on 20-4-2015 by Mesa]
[Edited on 20-4-2015 by Mesa]
|
|
|
byko3y
National Hazard
   
Posts: 721
Registered: 16-3-2015
Member Is Offline
Mood: dooM
|
|
Water cooled condenser is a recipe for your disaster. It's okay to have all-borosilicate setup, until you don't have 310°C bumping acid gas separated
by 2 mm of glass from a 30°C water.
And once again, even if you are sure there would be no real danger to the glass by a thermal shock - you might crack the glass mechanically, thus few
drops of a cold water may come into contact with acid wapor, leading to rapid condensation sucking the water further into the flask filled with
boiling sulfuric acid - boom.
[Edited on 20-4-2015 by byko3y]
|
|
|
macckone
Dispenser of practical lab wisdom
    
Posts: 2168
Registered: 1-3-2013
Location: Over a mile high
Member Is Offline
Mood: Electrical
|
|
Traditionally lead or ceramic retorts were used with air cooling.
A good quality borosilicate retort might be better.
Just wish I had a source for a quartz or high temp ceramic one.
|
|
|
Deathunter88
National Hazard
   
Posts: 519
Registered: 20-2-2015
Location: Beijing, China
Member Is Offline
Mood: No Mood
|
|
Just found an online supplier in China who sells concentrated sulphuric acid drain cleaner. It look like reagent grade except it is just slightly
cloudy. Its the only one I know of that sells is in China.
Here: http://item.taobao.com/item.htm?spm=a1z09.2.9.257.giwjG2&...
Don't be discouraged by the dark brown colour in the photo. Mine looked completely white when it arrived.
PS. Wooo, 50th post 
[Edited on 21-4-2015 by Deathunter88]
|
|
|
Trizocy
Harmless

Posts: 34
Registered: 16-2-2015
Member Is Offline
Mood: No Mood
|
|
Thanks deathhunter88  ! !
|
|
|
wg48
National Hazard
   
Posts: 821
Registered: 21-11-2015
Member Is Offline
Mood: No Mood
|
|
I want to concentrate my 30% sulphuric acid. I don't want to waste any of it so I will use a 280mm Vigreux column in a all glass distillation set up.
I estimate the column is equivalent to 2 to 4 theoretical plates.
From the attached graph I estimate:
at 98% acid (boiling temp.) the vapour is 80% acid
at 80% acid (boiling temp.) the vapour is 4% acid
at 4% acid (boiling temp.) the vapour is <0.01% acid
Now I have to remove most of the water ie approximately 70% (water) of the original acid/water to reach 98%.
With two theoretical plates at the end approximately 4% of the distillate will be acid which on average is 2%(start to finish) of the 70% ie
<1.5% of the acid will be lost.
With three or more theoretical plates the loss of acid will be <1%. Starting from 1 litre I will produce at least 0.28 litres of 98% acid
Do I have above about correct?
Attachment: Sulfuric Acid & Water - Liquid Vapor Equilibrium Concentration.pdf (328kB)
This file has been downloaded 672 times
[Edited on 12-8-2016 by wg48]
|
|
|
Metacelsus
International Hazard
    
Posts: 2539
Registered: 26-12-2012
Location: Boston, MA
Member Is Offline
Mood: Double, double, toil and trouble
|
|
You should be aware that using a Vigreux column will make it take a lot longer.
|
|
|
wg48
National Hazard
   
Posts: 821
Registered: 21-11-2015
Member Is Offline
Mood: No Mood
|
|
Can you be a bit quantitative how many hours approximately?
[Edited on 12-8-2016 by wg48]
|
|
|
Deathunter88
National Hazard
   
Posts: 519
Registered: 20-2-2015
Location: Beijing, China
Member Is Offline
Mood: No Mood
|
|
I'd be willing to bet that little or none will be collected unless you insulate it very well. I have used a 300mm as an air cooled condenser for
distilling sulphuric acid and it does a good job of condensing most/all of the acid.
|
|
|
wg48
National Hazard
   
Posts: 821
Registered: 21-11-2015
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by Deathunter88  |
I'd be willing to bet that little or none will be collected unless you insulate it very well. I have used a 300mm as an air cooled condenser for
distilling sulphuric acid and it does a good job of condensing most/all of the acid. |
I expect to be collecting water with approximately 1% acid so the top of the column should be just over 100C. Yes I may well need insulation
presumable but not too much as I want condensation over the whole column or it will not be fractionating.
|
|
|
ave369
Eastern European Lady of Mad Science
   
Posts: 596
Registered: 8-7-2015
Location: No Location
Member Is Offline
Mood: No Mood
|
|
I have a personal method of sulfuric acid distillation. It involves two-step cooling: first you air-cool it in the "nose" of a retort. Second, you
water-cool it in a Liebig which is connected to the nose of the retort. Thus you can avoid thermal shock. Also, no horrible acid smoke: it all
condenses at 3-4 cm from the hot end of the Liebig.
[Edited on 25-8-2016 by ave369]
Smells like ammonia....
|
|
|
| Pages:
1
2 |