Anders Hoveland
Banned
Posts: 208
Registered: 15-6-2010
Member Is Offline
Mood: No Mood
|
|
New Energetic Compound Design
This compound should be relatively insensitive and powerful. It contains a furozan and a tetrazole joined by a diazo N-oxide.
The first step leaves off sort from the DADNE synthesis. Interesting use of protecting groups. 2 pyrazoles joined by a hydrazine, --NHNH--, for
instance, was characterized by researchers as powerful (7.4km/sec) and very stable (more than RDX) considering all the nitrogen contained. I forgot
to write in what one of the steps was, it uses concentrated trifluroacetic acid and H2O2 to do the oxidation.
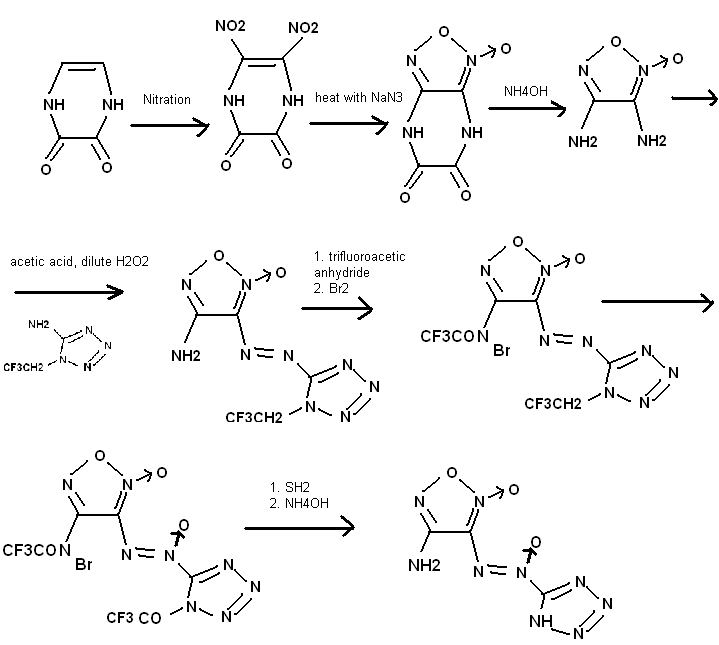
[Edited on 20-6-2010 by Anders Hoveland]
|
|
|
The_Davster
A pnictogen
      
Posts: 2861
Registered: 18-11-2003
Member Is Offline
Mood: .
|
|
1) No one designing new energetics considers 7400m/s powerful. For that you need to exceed RDX.
2) Diaminofuroxan was a synthetic target for a while. Turns out it decomposes on formation.
|
|
|
Anders Hoveland
Banned
Posts: 208
Registered: 15-6-2010
Member Is Offline
Mood: No Mood
|
|
In that case you could have two separate protecting groups on the amines, and only remove one of them. This should be stable, since amino, nitro
furoxan has been prepared. As long as one of the groups on the furoxan is electron withdrawing and can't ionize. That would mean there would have to
be two protecting groups on one of the amines, so there would be no H to ionie off and leave an electron on the nitrogen, that would move into the
furozan.
|
|
|
The_Davster
A pnictogen
      
Posts: 2861
Registered: 18-11-2003
Member Is Offline
Mood: .
|
|
The "protected" diaminofuroxan is 4-amino-3-azidocarbonylfuroxan. The amine can then form azo cpds as in the synthesis of
4,4'-dinitro-3,3'-azofuroxan (VOD >10km/s experimental). Heating in dioxane and water forms the amino from the azidocarbonyl species.
Aminonitrofuroxan is useless synthetically as preparation of either isomer spontaneously isomerizes to a mixture of 3-amino-4-nitro and
4-amino-3-nitro furoxan.
|
|
|
Anders Hoveland
Banned
Posts: 208
Registered: 15-6-2010
Member Is Offline
Mood: No Mood
|
|
Okay, this is all way to complicated for this type of forum. Lets simplify. Will analine and N-hydroxyl analine (PhNHOH) react together with H2O2 to
make diphenyl diazo N-oxide (PhNN(O)Ph)?
Possibly Aminotetrazole and Hydroxylaminotetrazole, with their a protecting group on one of the nitrogen in both their rings, would react similarly.
The compound, tetrazole-NHNH-tetrazole is well characterized and much stable.
Or just use
CAS Registry Number: 74999-19-2
5,5'-(1,2-hydrazinediyl)bis-2H-tetrazole,
then use trifluoroacetic anhydride to add protecting groups in place of the H's. Then oxidize the cetral diazo with concentrated H2O2 and conc.
trifluoroacetic acid.
Then get rid of the protecting groups by hydrolyzing with NH4OH. The result should be the two terazoles joined by a diazo N-oxide.
[Edited on 25-6-2010 by Anders Hoveland]
|
|
|