ChemichaelRXN
Hazard to Others
  
Posts: 103
Registered: 7-10-2010
Member Is Offline
Mood: Universal Eye
|
|
Benzoyl chloride from benzoic acid
What is your experience on this reaction?
With good ventilation, I was wondering what would occur if the benzoic acid was added to this reaction below. Would this be a suitable way to produce
and distill off benzoyl chloride?
SnO2 + 4 HCl → (( Cl2 )) + SnCl2 + 2 H2O
(Edited; Sorry this is what would form: SnO2 + 6 HCl → H2SnCl6 + 2 H2O
I was thinking a reaction that forms chlorine like; MnO2 + HCl —> MnCl2 + Cl2 + H2O )
Thanks,
ChemichaelRXN
[Edited on 9-12-2020 by ChemichaelRXN]
You are the same perception looking out, from the same elements around the universe.
You are everything to be anything to begin with.
Https://you-are.space
Https://syntharise.com
|
|
|
Boffis
International Hazard
    
Posts: 1847
Registered: 1-5-2011
Member Is Offline
Mood: No Mood
|
|
? what has tin got to do with benzoyl chloride? Your tin equation is garbage too. Anhydrous stannic oxide is not attached by hydrochloric acid; in
fact the reaction goes the other way!!
|
|
|
ChemichaelRXN
Hazard to Others
  
Posts: 103
Registered: 7-10-2010
Member Is Offline
Mood: Universal Eye
|
|
I am a laid back person, so you can just see how you sound...
Germanium, tin and lead oxides
For example, they all react with hydrochloric acid. This can be summarised as: For example, with tin(II) oxide, you get tin(II) chloride and water.
You also apparently get chlorine gas from the tin oxide reaction with hydrochloric acid.
I was thinking of utilizing the chlorine to react with benzoic acid and form benzoyl chloride that you can distill off.
Does anyone know this may work? I encourage creative, innovative thinking.
You are the same perception looking out, from the same elements around the universe.
You are everything to be anything to begin with.
Https://you-are.space
Https://syntharise.com
|
|
|
clearly_not_atara
International Hazard
    
Posts: 2723
Registered: 3-11-2013
Member Is Offline
Mood: Big
|
|
Boffis is an excellent chemist, you should listen to him.
Your post contains so many misconceptions it is hard to separate them from each other. In your first post you write SnO2, tin (IV) oxide, but in your
second post you mention tin (II) oxide. Neither efficiently produces chlorine by rxn with HCl; you may be thinking instead of MnO2 which gives this
rxn. Furthermore chlorine gas cannot convert carboxylic acids to the chlorides. Even if it could, benzoyl chloride cannot be produced in aqueous media
due to competing decomposition. Even if it could, the kinetics are highly unfavorable since chlorine would simply escape before it could react. And
even they were not, any attempt at distillation would obviously destroy the reactive product BzCl.
In short, this is so far from practical chemistry it might as well be knitting.
|
|
|
ChemichaelRXN
Hazard to Others
  
Posts: 103
Registered: 7-10-2010
Member Is Offline
Mood: Universal Eye
|
|
You can distill benzoyl chloride.
I was talking about SnO2. I must have been misinformed about the chlorine production. That seems how the equation lays out, but like you said it may
be MnO2 instead of SnO2 that would produce chlorine. MnO2 + HCl —> MnCl2 + Cl2 + H2O
[Edited on 9-12-2020 by ChemichaelRXN]
You are the same perception looking out, from the same elements around the universe.
You are everything to be anything to begin with.
Https://you-are.space
Https://syntharise.com
|
|
|
ChemichaelRXN
Hazard to Others
  
Posts: 103
Registered: 7-10-2010
Member Is Offline
Mood: Universal Eye
|
|
Here is knitting for you. Those who dare to think outside of the box always seem to get ridiculed.
https://www.prepchem.com/synthesis-of-benzoyl-chloride/
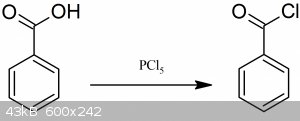
Benzoyl chloride could be prepared by reacting thionyl chloride or phosphorus pentachloride with benzoic acid.
50 grams of dry benzoic acid are treated in a 500 ml flask, with 90 grams of finely pulverized phosphorus pentachloride. The mixture is mixed well,
upon which, after a short time, the reaction takes place with an energetic evolution of hydrochloric acid, and the reaction mass becomes liquid.
During the reaction a lot of heat is released. After standing a short time, the completely liquid mixture is twice fractionated by collecting fraction
which boils at ~ 200° C, yielding 90 % of benzoyl chloride.
[ I was looking for another way. ]
Maybe the chlorine generated from the MnO2 and HCl would be better suited for benzaldehyde for the formation of benzoyl chloride. Benzaldehyde is
miscible with water slightly. Anyway, just thought I’d ask.
[Edited on 9-12-2020 by ChemichaelRXN]
You are the same perception looking out, from the same elements around the universe.
You are everything to be anything to begin with.
Https://you-are.space
Https://syntharise.com
|
|
|
Pumukli
National Hazard
   
Posts: 691
Registered: 2-3-2014
Location: EU
Member Is Offline
Mood: No Mood
|
|
I'm not sure I want to "weigh in" really but clearly_not_atara politely reminded you that Boffis is someone here who actually earned good reputation
because of his superb work! If he says something regarding a reaction (plan) then it is worth considering to say the least... Now it is starting to
feel embarassing but I can tell you the same things about clearly_not_atara too! If he says something regarding chemistry then you can trust it. :-)
I can feel your desire to "think out of the box", I myself like to do this from time to time but there is a difference between "thinking outside the
box" and "presenting superficially thought-out ideas".
Regarding your original idea about generating chlorine and using that to prepare benzoyl-chloride from benzoic acid you can be assured that
benzoyl-chloride (acid chlorides in general) are soo important reagents that their synthesis is thoroughly studied from lab-to-reactor scale and back!
If there was a better route (what was amateur friendly too) then we'd already use it.
Anyways, from benzoic acid to benzoyl chloride you can use the following reagents: PCl5, PCl3, SOCl2, Oxalyl-chloride, TCT. There are probably a
handful of other reagents too, in which I'm not 100% sure though. I can think of compounds like S2Cl2 or SCl2, or SiCl4, or SO2Cl2 or a few other
inorganic chlorides BUT elemental chlorine is not among them, be it generated from SnO2, MnO2, or any other way. So the basic flaw in your concept is
this last tidbit: Cl2 can not be used directly, you must first convert it to another form (PCl5, PCl3, etc) to be usable.
|
|
|
teodor
National Hazard
   
Posts: 874
Registered: 28-6-2019
Location: Heerenveen
Member Is Offline
|
|
This is known not to be happen. As far as I know neither with hydrated form (which forms different compound) no with anhydrous SnO2 which, according
to some studies, can go in solution this way only under long reflux with HI. Practically we use a fusion with something to get something water-soluble
from this nice compound.
|
|
|
ChemichaelRXN
Hazard to Others
  
Posts: 103
Registered: 7-10-2010
Member Is Offline
Mood: Universal Eye
|
|
Ok, great, thank you for the info., I appreciate it!
You are the same perception looking out, from the same elements around the universe.
You are everything to be anything to begin with.
Https://you-are.space
Https://syntharise.com
|
|
|