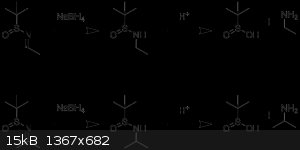
benzoyl - 17-8-2017 at 05:17
If you take a tert-butanesulfinyl ketimine, reduce it with NaBH4 and then cleave the suflinyl group via hydroylsis, is a primary or secondary amine
produced?
The original Ellman paper suggests a primary amine, but other sources indicate that tert-butanesulfinyl ketimines produce secondary amines and
tert-butanesulfinyl aldimines produce primary amines.
Sigmatropic - 17-8-2017 at 08:47
Ketimine --> primary amine with secondary carbon
Aldimine --> primary amine with primary carbon
see attached scheme.