
gmiguel - 13-5-2017 at 12:31
gmiguel - 13-5-2017 at 12:36
I'm interested in synthesis of the above molecule. The classic synthesis involves caustic fusion which creates considerable waste. I know this subject
was discussed on this discussion board previously.
I'm interested in any new ideas. Could an aryl sulfotransferase enzyme work?
Comments please.
CuReUS - 14-5-2017 at 01:39
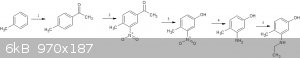
1.FC acylation of toluene - http://pubs.acs.org/doi/suppl/10.1021/ja8026899/suppl_file/j...
2.nitration of 4-methylacetophenone - http://pubs.acs.org/doi/abs/10.1021/ja1047736
3.One pot baever villager oxidation/hydrolysis on 4-methyl,3-nitroacetophenone -
http://www.ingentaconnect.com/contentone/stl/jcr/2000/000020...
4.reduction of 4-methyl,3-nitrophenol - http://www.sciencedirect.com/science/article/pii/S0960894X10...
5.ethylation of 4-methyl,3-aminophenol - http://onlinelibrary.wiley.com/doi/10.1002/jhet.5570260509/f...
The phenol can also be deprotected after the ethylation.(it might get simultanously deprotected during the ethylation  )
)

 )
)