RIPPER - 23-9-2004 at 03:57
keto nitrazines
I have a idea for energetic materials
with the O double-bound to the nitrazine-rings.
exambles:
triketo-RDX [(C=O)-(NNO2)]3->
1,3,5-trioxo-2,4,6-trinitro-2,4,6-triazine
tetraketo-HMX [(C=O)-(NNO2)]4->
1,3,5,7-tetraoxo-2,4,6,8-tetranitro-2,4,6,8-tetrazine
or
1-oxo-2,3-dinitro-2,3-diazine (C=O)-(NNO2)-(NNO2)->
and so on.

JohnWW - 23-9-2004 at 09:14
There is nothing on "keto-nitrazine" on Google. There were 950 responses for nitrazine, which is apparently used in medical chemistry as an
alkaline pH-determining reagent, especially in testing amniotic fluid, and also urine and saliva.
John W.
ketos
RIPPER - 27-9-2004 at 04:31
keto = one
nitrazine = nitro aza
or nitro azine
examble:
RDX-trione
(Synth. of isocyanuric trichloride)



New Ideas
RIPPER - 1-10-2004 at 04:07
Some new ideas to energetic materials.
RDX-trioxide
2,4,6-trinitro-2,4,6-triazacyclohexane-1,3,5-trioxide
trioxinetrinitrate
2,4,6-trinitrotrioxine
trinitrocyclohexanetrioxide
2,4,6-triamino-2,4,6-trinitro-1,3,5-trioxine
triaminotrinitrotrioxine
triaminotrinitrocyclohexanetrioxide



Jome - 1-10-2004 at 16:10
By adding oxygens (how are you going to oxidise the aromatic rings?) you're adding weight without increasing the energy output.
Replys
jacKy - 3-11-2004 at 05:15
I`ve found Sites at the E & W Forum
that writing gabbish to RIPPERS theoretical topic here !
nitrazine derivates
Whats your oppinion ?

David Marx - 3-11-2004 at 09:19
What does the last post even mean? "gabbish"?
The level of grammar has dropped dramatically on this site recently. Especially with all these schizophrenic posters!
sylla - 5-11-2004 at 09:10
There is too much oxygen in triketo-rdx and this would probably give bad results (as an explosive) but monoketo-rdx seems to be very nice 
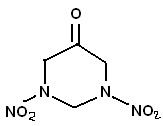
Reference - 17-7-2010 at 22:46
A little off, topic, but I was reading about dihydrosxyacetone, and its condensation with amines. "The pigment is the product of reactions between
dihydroxyacetone (DHA) and amino acids". I was wondering if maybe dihydroxyacetone would condense with the methylene dinitramine (MEDINA) ? Making
something like in the picture. Can't think of a use for it, but would probably be the most stable keto-RDX version.