

Quote: Originally posted by Poppy  |
Quote: Originally posted by bfesser  |

Quote: Originally posted by woelen  |
Quote: Originally posted by kmno4  |
Quote: Originally posted by bfesser  |
Quote: Originally posted by blogfast25  |
Quote: Originally posted by blogfast25  |
Quote: Originally posted by adamsium  |
 I donated it to Wikipedia. I
made it myself some years ago. I still have these ampoules, they are very nice demo objects and sometimes I show them to friends and show the
differences under different types of light.
I donated it to Wikipedia. I
made it myself some years ago. I still have these ampoules, they are very nice demo objects and sometimes I show them to friends and show the
differences under different types of light.Quote: Originally posted by adamsium  |
 !!! - and dissolve again). Anyway, the hydroxides seems stable as I lately added
about 5mL conc. sulfuric acid to this solution and that didn't dissolve the white ppt. seen.
!!! - and dissolve again). Anyway, the hydroxides seems stable as I lately added
about 5mL conc. sulfuric acid to this solution and that didn't dissolve the white ppt. seen.
Quote: Originally posted by woelen  |
Quote: Originally posted by watson.fawkes  |

Quote: Originally posted by watson.fawkes  |
Quote: Originally posted by blogfast25  |
Quote: Originally posted by woelen  |
Quote: Originally posted by bfesser  |
Quote: Originally posted by bfesser  |
Quote: Originally posted by blogfast25  |

Quote: Originally posted by woelen  |
Quote: Originally posted by Bezaleel  |
Quote: Originally posted by blogfast25  |
Quote: Originally posted by Bezaleel  |

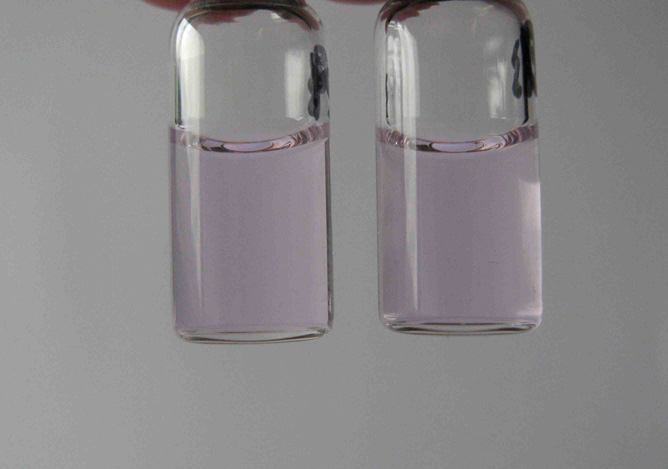
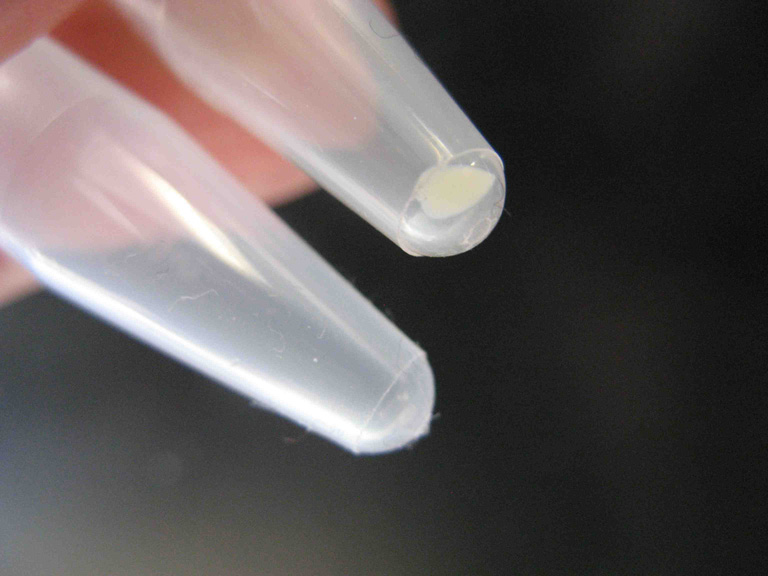

Quote: Originally posted by Bezaleel  |
Quote: Originally posted by blogfast25  |
Quote: Originally posted by watson.fawkes  |
Quote: Originally posted by blogfast25  |
Quote: Originally posted by Poppy  |
Quote: Originally posted by phlogiston  |
Quote: Originally posted by Poppy  |