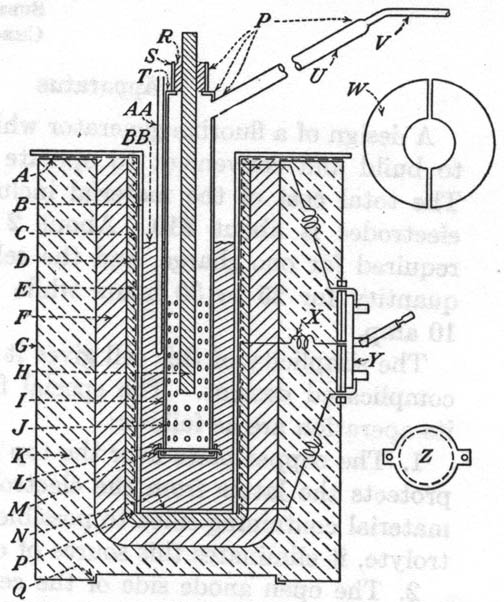
| A, copper plate 1/8 in. thick of sufficient size to overlap the furnace. It is securely silver soldered to the well. The negative
lead from the direct-current source is connected to this plate by means of a battery clip; B, well, made of copper tubing 12 in.
long, 3 in. i.d., and a wall thickness of 1/8 in.; C, asbestos paper wrapping; D, nichrome wire heating element, 50
ft. of No. 18; E, alundum cement coating about 1/4 in. thick; F, coating of a mixture of fire clay and sand about 1
in. thick. This gives rigidity and permanence to the heating unit and is inexpensive; G, sheet-metal can, usual type of container
for chemicals; H, electrode, graphite rod 12 in. long and 1/2 in. in diameter, made of a special mix with a minimum of silicon
binder. The positive lead from the direct-current source is connected to the top of this electrode by means of a battery clip; I,
diaphragm, made of copper tubing 12 in. long, 1 3/4 in. in diameter, and with a wall thickness 1/16 in.; J, holes drilled in
diaphragm, 1/8 in. in diameter; K, magnesia or asbestos refactory packing; L, bottom of diaphragm made of 1/16-in.
copper sheet; M, copper wire for connecting the bottom to diaphragm; N, bottom of well, made of 1/8-in. copper
sheet; P, joints made with hard-silver solder; Q, press lid of sheet-metal can; R, Portland cement
seal; S, seal tube made of copper tubing 1 1/4 in. long, 1 in. in diameter, and 1/16-in. wall size. It is convenient to support the
diaphragm by means of a clam on this tube; T, copper disc with 3/4-in. center hole; U, fluorine exit tube, 1/2 in.
in diameter and 12 in. long; V, copper tube 1/4 in. in diameter. W, cover for cell when not in use, made of
1/16-in. copper plate in two parts with center hole large enough to fit around diaphragm; X, center lead from heating element;
Y, double-throw double-pole switch, so connected as to enable the two halves of the heating element to be placed in either parallel
series. The series connection is for the regular operation of the cell; the parallel, for raising the temperature. Transite boards inside and
outside the metal can form the base upon which the switch is fastended; Z, detail construction of bottom of diaphram;
AA, thermometer well, a copper tube sealed at the bottom. It is convenient to have this only temporarily fastened to diaphragm with
copper wire or better supported from a ring stand. A thermocouple is more convenient to use then a mercury thermometer. If the later is used, it
should be covered with a film of heavy oil; BB, electrolyte. |

 . Is Fluorine not very reactive against copper?? I always thought only
Nickel-lined vessels could resist Fluorine...but if not, this would be very interesting and useful. Surely we could adapt this to a new, modified
cell that does not use asbestos.
. Is Fluorine not very reactive against copper?? I always thought only
Nickel-lined vessels could resist Fluorine...but if not, this would be very interesting and useful. Surely we could adapt this to a new, modified
cell that does not use asbestos.
 )
)


