Difference between revisions of "Benzocaine"
From Sciencemadness Wiki
m |
m |
||
| Line 52: | Line 52: | ||
| AtmosphericOHRateConstant = | | AtmosphericOHRateConstant = | ||
| Appearance = | | Appearance = | ||
| − | | BoilingPt = | + | | BoilingPt = 310 °C |
| BoilingPtC = | | BoilingPtC = | ||
| BoilingPt_ref = | | BoilingPt_ref = | ||
| Line 68: | Line 68: | ||
| pKb = | | pKb = | ||
| Solubility = slightly soluble in water. | | Solubility = slightly soluble in water. | ||
| − | |||
| Solvent = ethanol, chloroform, ethyl dilute acids. | | Solvent = ethanol, chloroform, ethyl dilute acids. | ||
| VaporPressure = | | VaporPressure = | ||
Revision as of 11:38, 27 January 2016
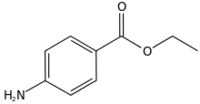
| |
| Names | |
|---|---|
| IUPAC name
Ethyl 4-aminobenzoate
| |
| Systematic IUPAC name
Ethyl 4-aminobenzoate | |
| Properties | |
| C 9 H 11 NO 2 | |
| Molar mass | 165.189 g/mol |
| Density | 1.17g/cm3 |
| Melting point | 89 °C |
| Boiling point | 310 °C |
| slightly soluble in water. | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
(Introduction)
Contents
Properties
Chemical
Benzocaine is a local anesthetic. It is the ethyl ester of PABA or ParaAminoBenzoic Acid.
Physical
Availability
Delete this section if not applicable
Preparation
Delete this section if not applicable